Theoretical studies replace double bonds with dative in hypervalent iodine compounds
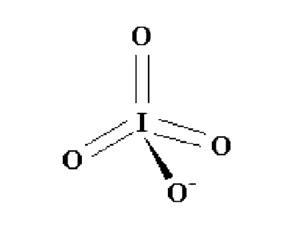
Models contend iodine double bonds was first published by Chemistry World.
Theoretical studies replace double bonds with dative in hypervalent iodine compounds

Models contend iodine double bonds was first published by Chemistry World.
No comments yet