Stephen Ashworth shows you how to purify a mixture with just a little evaporation in this simple experiment from our ‘Avogadro’s lab’ series
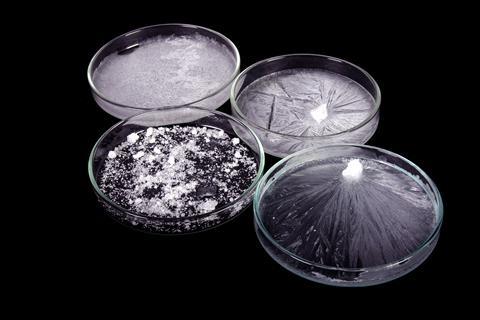
Crystals start to form when a material in a solution is no longer soluble in the surrounding solvent. This might be because the solution has cooled, or solvent has evaporated making the solution more concentrated. If crystallisation happens quickly, the crystals formed tend to be small. You may have already found this if you have tried the Global Experiment. Slow crystallisation allows larger crystals to form.
Crystals start to form when a material in a solution is no longer soluble in the surrounding solvent. This might be because the solution has cooled, or solvent has evaporated making the solution more concentrated. If crystallisation happens quickly, the crystals formed tend to be small. You may have already found this if you have tried the Global Experiment (http://rsc.li/1u5VQCQ). Slow crystallisation allows larger crystals to form.
Igneous rocks (such as basalt and granite) contain some of the most striking examples of natural crystals, which are created by a process called fractional crystallisation. These rocks are made up of complex mixtures of minerals from the Earth’s mantle. When this molten rock cools, the minerals in the mixture crystallise at different rates, depending on the composition of the mixture and the rate of cooling. This is why such rocks have crystals of pure materials embedded in them.
Fractional crystallisation is a powerful purification technique. The same process that defines the structure of rocks can be used to separate and purify dissolved solids.
If the substances in a mixed solution have sufficiently different solubilities (this is defined as the maximum amount of solid that can be dissolved in a volume of solvent), they can be separated from the mixture by fractional crystallisation. This experiment, which you can try, demonstrates the principle of how the process works.
Try it yourself
Equipment required
You will need:
- Table salt
- Sugar
- Water
- A measuring jug
- A shallow dish
Procedure
The solubility of salt in water is lower than that of sugar, so when they are mixed in solution the salt will tend to crystallise first.
Mix three teaspoons of salt and three teaspoons of sugar in 100 cm3 of water. When they are dissolved you can start crystallisation by warming the water or just allowing it to evaporate slowly from a shallow dish.
You can analyse the solution by taste because we know that salt, sugar and water are not toxic. To begin with the solution tastes predominantly of salt. As the water evaporates and the salt crystallises out of the solution, the sugar becomes more concentrated and the liquid will start to taste sweeter. Quite a lot of water might have to evaporate before the change is very noticeable, though.
Here we are using about the same mass of both substances, but as sugar is almost ten times more soluble than salt, the crystals we make are high purity salt. This means that the remaining liquid is a concentrated sugar solution containing the remaining uncrystallised salt.
Complex mixtures
Fractional crystallisation will also work for much more complex mixtures. In many cases varying the temperature is used to alter a substance’s solubility so that crystals form as the solution cools. For example, concentrated fruit juices and other heat sensitive liquids are often made in this way. However, heating juice to evaporate water can spoil the flavour. Instead juice is cooled and the solid that forms at low temperature is mainly water ice containing a low concentration of other juice components. When the solid is removed, concentrated juice is left behind.
So next time you drink fruit juice (made from concentrate) or see crystals embedded in rock samples, what you see (or drink) are the results of fractional crystallisation.
Additional information
This article originally appeared in The Mole, the student magazine published by the Royal Society of Chemistry from 2012 to 2015.









No comments yet