Chemical processes occurring in the atmosphere determine current and future atmospheric compositions, which in turn affect climate, air quality, and the spread of pollution. What are these processes and can they help us understand the likely impacts of climate change upon atmospheric conditions?
-
Atmospheric chemistry is dominated by gas phase radicals
-
Tropospheric levels of .OH control levels of greenhouse gases in the atmosphere

While the bulk of our atmosphere is made up of nitrogen, oxygen, carbon dioxide and water vapour, atmospheric chemistry is dominated by reactions of trace constituents, specifically gas phase radicals (species with unpaired electrons), present in parts per million (ppm), billion (ppb) or even trillion (1 part in 1012). Through chain reaction cycles, these highly reactive compounds control the levels of the more abundant species in the atmosphere.
Tropospheric chemistry

The basic chemistry of the first 10-15 km of our atmosphere, the troposphere, is dominated by the reactions of hydrocarbons with hydroxyl radicals (•OH), see Scheme 1. Ozone (O3) is broken down by sunlight, leading to high-energy oxygen atoms, which may be stabilised by colliding with nitrogen or oxygen molecules, or may react with water vapour to form two •OH radicals. The •OH radicals then react with hydrocarbons to form organic peroxy radicals (RO2•) - for example, CH3O2• is formed when •OH reacts with methane.
What happens next depends upon the availability of the nitrogen oxide radicals, NO• and NO2• (collectively called 'NOx'). The main source of NOx is the combustion of fossil fuels - atmospheric scientists refer to low NOx environments, eg the remote atmosphere over the Pacific Ocean, as 'clean' and high NOx environments, eg cities, as 'polluted'. If NOx levels are low, the peroxy radicals react with HO2• to form peroxides, such as methylperoxide (CH3OOH) and hydrogen peroxide (H2O2). The peroxides are relatively stable and soluble in water, leading to their removal through precipitation, and represent the end of the reaction chain in clean environments.
However, in the presence of NOxfurther reactions occur: NO• reacts first with the organic peroxy radicals, forming HO2•, NO2• and an aldehyde molecule, and then also with HO2•, forming •OH and NO2• again. Thus, in the presence of NOx, hydrocarbons are oxidised as part of a propagating chain, with the initial formation of a single •OH, leading to the oxidation of several molecules of, for example, CH4. The fate of the NO2• formed in the process is critical: NO2• readily undergoes photolysis in the atmosphere to form oxygen atoms and NO•. The oxygen atoms react with molecular dioxygen (O2), forming ozone (O3) - for each molecule of methane oxidised by •OH, two molecules of O3 are formed (Scheme 1), and for each molecule of ozone photolysed to initiate the chemistry, many more can be generated by the resulting cycle. Competing with the cycle (chain propagation) are the radical removal (chain termination) steps of peroxide formation and the reaction of •OH with NO2• to form nitric acid.
Impacts of tropospheric chemistry
The chemical processes shown in Scheme 1 control many aspects of atmospheric composition: reaction with •OH dominates the removal of most pollutants emitted to the atmosphere, determining their lifetime and abundance. Tropospheric •OH levels thus regulate the self-cleansing ability, or oxidising capacity, of the atmosphere, and control the levels of greenhouse gases such as CH4. (Note CH4is the second most significant greenhouse gas after CO2; CO2 does not react with •OH.)
This behaviour has been used advantageously in the case of hydrochlorofluorocarbons (HCFCs), such as CHF2Cl, the hydrogenated replacements for chlorofluoro-carbons (CFCs, such as CF2Cl2), introduced in the 1980s and 1990s to prevent stratospheric ozone loss. The •OH reacts with HCFCs by removing the H atom to form water, leaving an alkyl radical, which then undergoes further breakdown in the atmosphere. So HCFCs are removed in the lower atmosphere, while CFCs pass unimpeded to the stratosphere.1
Tropospheric chemistry also affects air quality. Oxidation of hydrocarbons such as methane, carbon monoxide and larger molecules such as volatile organic compounds (VOCs) in the presence of NOx leads to the production of ozone, see Scheme 1. This is important because ozone is both a greenhouse gas, contributing to climate change, and a major constituent of the photochemical smog observed over many cities during summertime, which is harmful to human and animal health, vegetation and certain materials such as plastics.
Current UK National Air Quality strategy suggests a target level of 50 ppb for ozone. Mild health problems, owing to ozone inhalation may become noticeable for sensitive individuals at levels of 50 - 90 ppb, requiring them to spend less time outdoors, while at levels over 90-100 ppb asthma sufferers may need to increase their use of inhaler.2 For comparison, ozone levels in remote environments may be 5-10 ppb, typical background UK levels are around 30 ppb, while levels in Los Angeles in the late 1970s reached up to 500 ppb.3 Thus by understanding tropospheric atmospheric chemistry we can put forward strategies to improve air quality, such as identifying particular compounds with a high ozone creation potential, whose emissions should be controlled.
Simulation chambers and field experiments

Our understanding of atmospheric chemistry has been developed through a combination of laboratory studies, field measurements and computer modelling. Laboratory experiments are used to determine the molecular properties (reaction rate constants and photolysis rates) governing the fate of individual atmospheric species. The results are used to design reaction mechanisms for specific species, which are then used in computer models of atmospheric chemistry. Comparison of such models with field measurements of concentrations in the ambient atmosphere are then used to test the model, and refine the reaction mechanism as necessary.
'Smog chambers' or simulation chambers are used to study artificial atmospheres. The EUPHORE ('European Photoreactor') simulation chamber in Valencia, Spain, for example, comprises two 200 m3 hemispherical reaction vessels, each ca 4 m tall, made from a (transparent) non-reactive polymer 'bag' 0.2 mm thick. Each vessel has covers which can be raised to block sunlight, and houses a range of analytical instruments. Artificial atmospheric mixtures of a particular chemical system are introduced into the chambers and the photochemical reactions started by opening the covers and admitting sunlight. The resulting changing chemical composition is monitored.
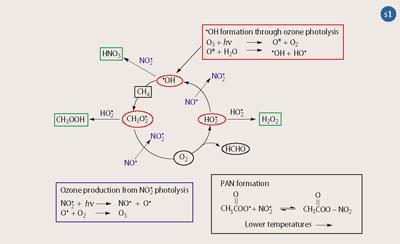
Figure 1 shows concentrations of benzene, ozone and NOx (black lines) during an experiment to investigate benzene removal. This was obtained as part of the EXACT (Effects of the Oxidation of Aromatic Compounds in the Troposphere) research project involving a consortium of scientists from European universities.4 The graphs show the concentration of benzene falling because of its reaction with •OH, and the resulting increase in ozone levels through the reactions shown in Scheme 1. Also shown in the figure are the predictions (red lines) of an atmospheric model, the Master Chemical Mechanism,5 which agree well with the observations, giving us confidence in the reaction mechanisms.
Another approach involves taking a range of measurements of as many of the atmospheric reactants as possible at a particular location over a short time, typically 4-6 weeks. This often involves teams of scientists from different institutions, specialising in a variety of measurement techniques. The data acquired are then compared with calculations and model predictions. Consider, for example, •OH, one of the most highly reactive radicals. The lifetime of •OH is extremely short, typically 0.1-2 s, in the lower atmosphere. Thus •OH concentrations are determined by local chemical production and loss, rather than transport, ie •OH is not blown to a particular location from elsewhere. This behaviour allows us to use observations of very short lived species like •OH to test our understanding of the chemistry. We suggest a chemical mechanism based upon our best knowledge, measure the levels of (comparatively) long-lived species such as O3, NOx and VOCs, use the mechanism to calculate the levels of short-lived species based upon measurements of those with longer lifetimes, and compare the resulting calculations with measurements of •OH etc. This approach eliminates the need to worry about the transport of atmospheric constituents by winds.
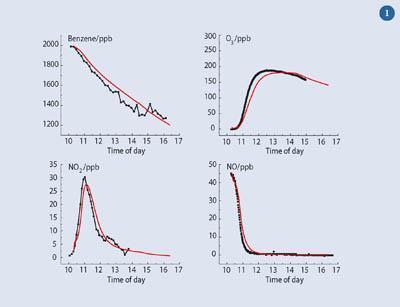
Figure 2 shows the results of measurements of HO2• radicals at a site in Mace Head on the west coast of Ireland - samples comprised clean marine air from the Atlantic Ocean - together with simulations of their concentrations. The observed data (green/yellow dots) were obtained during the NAMBLEX (North Atlantic Marine Boundary Layer Experiment) operation, August 2002.6 The radical levels rise and fall over a 24-hour period, reflecting the role of sunlight in their generation, with HO2• levels peaking at 4-6 pptv (indicating the very low levels of atmospheric radical species).
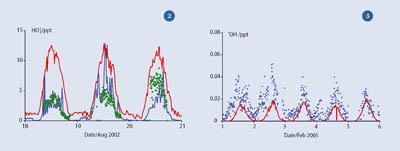
Figure 2 also shows the results from two models - the first (in red) used the 'standard' chemistry shown in Scheme 1, while the second (in blue) includes reactions of iodine oxides, produced by seaweeds and marine biota, which convert HO2• into •OH, thus lowering the HO2• level.7 The more comprehensive model, including the iodine chemistry, clearly does a better job of reproducing the observations.
Many atmospheric chemists are currently trying to assess the significance of iodine chemistry. Is the iodine restricted to a few coastal sites with exposed seaweed beds at low tide, or are the iodine compounds found more widely over the open ocean?
Chemistry in a changing atmosphere

Atmospheric chemistry and climate are interrelated. Chemical processes determine the abundance of greenhouse gases such as methane and ozone, and also contribute to the production of particulate matter (aerosol) when the oxidation of organic species leads to the production of low volatility compounds. In turn, the effects of climate change are likely to lead to significant changes in the chemical processes occurring; these can be broadly divided into changes owing to the different physical environment (humidity, temperature), and changes in emissions to the atmosphere.
The rate of most atmospheric reactions vary with temperature, so warmer (average) temperatures will change the relative importance of the competing pathways in Scheme 1. For example, at around 290 K, the rate of the •OH + CH4 reaction increases by 2 per cent per degree Kelvin.8 One interesting consequence of this relates to the transport of nitrogen oxides from pollution sources to remote regions.
A major mechanism for this transport is through the formation of peroxy acetyl nitrate (PAN), following the reaction between NO2• and acetyl peroxy radicals (Scheme 1). PAN is in thermal equilibrium with NO2• and CH3C(O)OO•, with low temperatures favouring PAN. Consequently PAN can be formed in a (typically low altitude) polluted region, lofted to higher altitudes where temperatures are lower and the molecule is stable, and in this state be transported to remote locations 1000s of km from the pollution source.
If the air temperature then increases (for example, following descent to lower altitudes) PAN decomposes, releasing NOx into the clean environment (the decomposition rate increases by 17 per cent per K at 288 K). Higher temperatures will increase the rate of thermal decomposition of PAN, and thus reduce its ability to transport NOx. All other factors being equal, this change will lead to increased NOx levels close to emission sources, and reduced NOx in remote areas.
Predicted global warming is likely to lead to an increase in atmospheric water vapour levels. On its own, this effect will lead to increases in the rate of production of •OH, as a larger fraction of the excited oxygen atoms formed from ozone photolysis react with H2O rather than with N2 or O2. However, atmospheric levels of VOCs, particularly CH4 and CO, are also predicted to increase, which will reduce •OH levels (because they are the main sinks, or reaction partners, for •OH). Overall, model simulations indicate that the latter effect will dominate, and tropospheric •OH levels may decrease by 6-25 per cent by 2100.9 Reduced •OH concentrations will lead to reduced rates of removal of pollutants and global warming gases such as methane (an example of a positive climate feedback loop).
Globally, emissions of VOCs and NOx are expected to increase over the period to 2100, both through human activities (particularly increased urbanisation) and natural biological processes. Certain kinds of plants and trees, for example, emit VOCs, and respond to increased temperatures with enhanced emissions of hydrocarbons such as isoprene (C5H8). This will lead to increased ozone production through the reactions shown in Scheme 1.
An insight into the conditions which might become more commonplace was provided by the heatwave over Western Europe during the first two weeks of August 2003. An extended period with temperatures reaching the upper-30s (°C) occurred together with stable weather conditions. This led to prolonged chemical processing of polluted air from major urban centres, and high ozone levels reaching over 110 ppb at a number of locations across south-east England.2 An estimated 200-500 deaths were brought forward, owing to the increased ozone levels alone during this period.10
Model studies based upon our current understanding of atmospheric chemistry, and estimates of likely future emissions under various scenarios, predict the change in tropospheric ozone levels which might be expected in the future. Surface ozone levels at mid-latitudes are predicted to rise by approximately 30 ppb by 210011 - an increase which will lead to background ozone levels being comparable to the limits set in current air quality standards. If these predictions are borne out, people's health in polluted regions will be adversely affected - and the improvements in air quality11 observed in many Western nations over the past few decades may be reversed (as far as ozone is concerned) in the future.
Clues from the ice
Predicting atmospheric chemistry in the future is difficult, not least because of the uncertainties in predicting future pollutant emissions, and understanding the atmosphere's natural variability, and natural response to changes in climate. Clues to the latter can be gleaned from the past, specifically from measurements of atmospheric gases trapped in air bubbles in snow which has been compressed into ice, and held undisturbed for thousands of years in the polar ice caps.
In addition to information about past temperatures and CO2 levels, air from ice cores can tell us how reactive the atmosphere was in the past - through levels of peroxides and nitrate, which are related to the concentrations of the oxidant radicals in Scheme 1. Recent research has, however, shown that the interaction between the atmosphere and the snow surface, under sunlight, can lead to a range of chemical effects. These include the conversion of nitric acid to more reactive forms, leading to release of NOxfrom the snow surface, and the production of organic compounds such as formaldehyde, both of which lead to enhanced levels of •OH and HO2•.12 One area of current research is to understand the mechanisms behind these processes, through field measurements of atmospheric and snowpack composition.
Figure 3 shows results from a site at the British Antarctic Survey's Halley Base, during the recent CHABLIS (Chemistry of the Antarctic Boundary Layer and the Interface with Snow) project, involving a team of scientists from UK universities. The •OH concentrations were recorded over a five-day period in January 2005 (blue dots), and simulated •OH concentrations (red line) were calculated assuming NOx levels were very low, as we would expect from such a remote, clean location. The actual data are higher, reflecting the snowpack emissions of NOx (driving the radical cycling shown in Scheme 1) and formaldehyde (acting as an additional HOx source).13
Our ongoing research aims to constrain the chemical processes occurring, so that we can understand the chemistry of our atmosphere above the polar ice sheets, and the link between atmospheric composition and the air trapped in ice cores. This, in turn, will continue to improve our understanding of atmospheric chemistry, and our ability to predict how the atmosphere will respond to future changes.
William Bloss is a lecturer in atmospheric science in the school of geography, Earth & environmental sciences at the University of Birmingham, Edgbaston, Birmingham B15 2TT.
Related Links
Air quality data and information
The Intergovernmental Panel on Climate Change
IPCC Fourth assessment report on climate change
The EXACT project
References
- World Meteorological Organisation (WMO) report no 47: Scientific assessment of ozone depletion, 2002.
- UK National Air Quality website
- R. P. Wayne, Chemistry of atmospheres. Oxford: OUP, 2000.
- The EXACT project website
- The master chemical mechanism website
- D. E. Heard et al, Atmos. Chem. Phys., 2006, 6 , 2241.
- W. J. Bloss et al, Geophys. Res. Lett., 2005, 32 .
- R. Atkinson et al, Atmos. Chem. Phys., 2004, 4 , 1461.
- Intergovernmental Panel on Climate Change (IPCC), Third assessment report: The scientific basis, 2001.
- J. R. Stedman, Atmos. Environ., 2004, 38, 1087.
- See D. Shallcross, Educ. Chem., 2006, 43 (5), 131.
- F. Dominé and P. B. Shepson, Science, 2002, 297, 1506.
- W. J. Bloss et al, Atmos. Chem. Phys. Discuss., 2007, 7 , 2893.






No comments yet