Conducting composites, based on metal- or carbon-filled polymers, have been a focus for research for the past 100 years. Ten years ago, however, a serendipitous discovery led to a new class of materials - quantum tunnelling composites (QTCs).
Quantum tunnelling composites (QTCs) were first made by accident in 1997 by David Lussey, a research engineer in Darlington who at the time ran a security business. Asked by a local FE college to solve its problem of students stealing computers, Lussey's solution involved attaching to the computers a tag which was wired to an alarm system. Removing the computer would detach the tag and trip the alarm. For this to work Lussey needed a conducting adhesive with which to stick the tag and complete the alarm circuit. 'No conducting adhesive available at the time offered the 'tack' properties [similar to the adhesive on Post-it notes] I wanted', Lussey told InfoChem. 'So I decide to make my own'.

Homework
Working in his laboratory at home, Lussey made hundreds of formulations by mixing different combinations of metal particles and adhesives in a poly(ethene) pestle and mortar. To test each formulation he used the material to glue together two pieces of aluminium and then measured the resistance across the contact using an ohmmeter.
Among the many materials he made, one formulation, consisting of fine nickel particles and silicone, exhibited unusual properties. At first his test showed that the material was an insulator and therefore useless as a conductive adhesive. However, when Lussey tried to pull the aluminium plates apart he noticed a reading on the meter - the conductivity had increased.
A few weeks later Lussey returned to his strange material and decided to set a sample in a mould. The result was a black, rubber-like solid, similar in feel and texture to car tyre rubber. He took the sample to physicist Professor David Bloor at Durham University to confirm his initial findings and find out more about its properties.
Bloor's tests showed that in its 'relaxed' state the composite behaved like an insulator with resistance of 1014 Ω. But when deformed, ie squeezed, stretched or twisted, its resistance fell off exponentially to less than 1 Ω, making the composite a metal-like conductor. Analysis of the sample's composition using scanning electron microscopy (SEM) revealed individual micron-sized (10-6 m) nickel particles dispersed in the silicone polymer. Importantly, the SEM images showed that the surfaces of the nickel particles were uneven with nanoscale (10-9 m) spikes.
Shape of things to come

In conventional conducting composites a polymer matrix is loaded with particles packed close together, eg high-density poly(ethene) and carbon black particles. 'These are made on an industrial scale by mixing the components for long periods using a machine blender', explains Lussey. 'This offers no control over the shear forces caused in the blend by the mixing process. As a result the particles rub against one another until they are smooth and round'. When the composite material is squeezed neighbouring carbon particles touch each other, creating conduction pathways which lower the resistance of the material. This conduction process is called percolation.
Lussey, working in collaboration with Bloor, discovered that the nanoscale spikes on the surface of the nickel particles in his composite are key to its unique conducting properties. By gently mixing the materials by hand, he had limited shear forces during mixing and so maintained the particles spiky shape. Although packed close together, the nickel particles always remain separated by the silicone polymer even when the material is deformed. Electronic charge on the particles is concentrated onto the tips of the spikes on the surface, which generates high charge densities. When the composite is deformed the particles are brought close enough together for electrons from these areas of high charge density to 'jump' from one particle to another other. This process is known as quantum tunnelling.
Lussey realised that his QTC's unique resistance response to changes in inter-particle spacing, changing from insulator ('off') to conductor ('on'), could have potential applications in switching or touch/pressure sensitive electronic devices. In 1997 he started up Peratech, a company to develop more QTCs and find applications for this new technology. Using a patented manufacturing process, Peratech can now make QTCs from almost any conducting or semi-conducting metal and a wide variety of polymers.
Switch on to QTCs
Switches, keypads and joystick controls, which rely on two metal surfaces to make an electrical contact when pushed together, are just some of the applications where QTCs can offer a more durable alternative. There are others.
The Shadow Robot Company, a London-based engineering firm, uses QTCs as pressure/force sensors in its tactile sensor for robotic hands. Fitted to a robotic hand as a 'skin', the dramatic response of QTCs to the slightest change in pressure provides a robot with 'first touch' sensitivity so that it can detect if it is holding an egg or a metal ball, if an object is slipping from its grasp, or if it has correctly fitted items together.
QTCs have applications in cars. For example, QTC sensors under car seats can detect whether an adult, child or baby is occupying a seat. In the event of a crash, this signal determines with what force airbags are deployed. In a similar way QTC bumper sensors can detect if a car has hit a pedestrian or a pillar. Having detected a collision with a pedestrian, the sensor sends a signal to trigger the bonnet to push up slightly. By doing this the bonnet can deform more, absorbing more energy in the collision, and dramatically reduce pedestrian head injuries.
A current focus of Peratech is developing QTCs for use in 'electronic noses' designed to detect volatile organic compounds (VOCs). Polymers in the presence of VOCs tend to swell as VOC molecules absorb at the surface. 'QTCs are so sensitive to changes in the spacing between the conducting particles in the matrix that swelling caused by VOCs at parts per million (ppm) concentrations induces a measurable change in the material's conductivity', explains Lussey. By choosing carefully polymers for its composites, Peratech is developing QTCs to 'sniff out' particular VOCs.
Being flexible like rubber, QTCs can be coated onto fabrics to make 'intelligent textiles'. Softswitch, a company owned by Peratech, produce QTC-based electronic fabrics. Some of these have been incorporated into a snowboard jacket that offers wearable controls for an iPod or mp3 player connected through a pocket.
Shrinking the third dimension
By altering the polymers used in QTCs, from silicones to aqueous- or organic-based polymers, films as thin as 10-5 m can be produced. These printable QTC films can be designed as a series of buttons on a keyboard. Future developments in this area could have a huge impact on many everyday electrical products. For example, keypads based on QTC films could allow mobile phones of the future to have dimensions similar to a credit card. 'QTC thin films are an exciting area for the future because they offer a means of shrinking the third dimension [ie depth] of electronic products. This could be QTCs' biggest and most obvious impact on everyday life', says Lussey.
Quantum leaps
Quantum tunnelling is a quantum-mechanical property of electrons. Quantum mechanics was developed from Max Planck's (1858-1947) quantum theory and is used to describe the properties of atoms and molecules. In quantum mechanics an electron is regarded as a wave rather than a solid particle, where the wave describes the probability of finding the electron at a particular location.
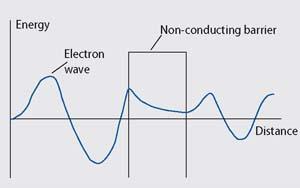
Electrical conduction is the flow of electrons through a material driven by an electric field. When a wave describing a conducting electron meets a barrier, eg the non-conducting polymer component of a QTC, the wave does not immediately drop to zero, but decays exponentially. This represents a decreasing probability of finding the electron in the insulator material. If the wave does not reach zero before it meets the other side of the barrier, it emerges on the other side. This implies there is a probability that the electron could be found on the other side of the barrier. So in effect the electron can 'tunnel' through the non-conducting barrier.
This article was originally published in The Mole









No comments yet