Here’s the solution to concentrating learners’ minds on the key aspects of this crucial topic
Solutions are all around us; in sweetened drinks, in the sea we swim in and sail on, and in everyday household cleaning products. But how are they formed and what do they look like on a particle level?
A solution is the homogeneous mixture formed when one substance (the solute) is uniformly distributed within another (the solvent). At 14–16 the term mainly applies to solid–liquid solutions, such as salt dissolved in water, but the term could equally be applied to the air we breathe (a gas–gas solution), carbonated drinks (gas–liquid solutions) and alloys (solid–solid solutions).
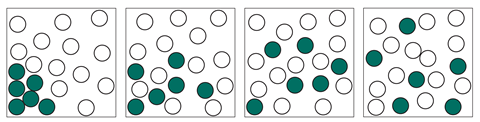
Younger students will have met the concept of a solute dissolving in a solvent to form a solution. They learn that dissolving is a physical change in which a solid breaks into smaller particles that spread out through the solvent.
At 14–16, introduce students to the idea of concentration. Then build on this with the concept of pH, quantitative chemistry calculations that involve one or more solutions, and investigating factors that affect reaction rate.
What students need to know
At 14–16 students need to know that:
- The relative solubilities of elements or compounds in water is affected by the bonding.
- The solubility of a particular solute is defined as the maximum mass of solute that will dissolve in 100 g of water at a given temperature.
- The concentration of a solution is the amount of solute dissolved in a defined volume, typically given in units of g dm-3 or mol dm-3.
- Crystallisation is used to produce solid crystals from a solution by evaporation and cooling.

What you need to know
The solubility of a particular solute in a solvent depends on the bonding in both the solute and the solvent. In general, polar solutes dissolve in polar solvents and non-polar solutes dissolve in non-polar solvents. Introduce this concept when looking at the solubility of ionic and covalent compounds in water.
Ionic compounds dissolve in water because water is polar and it forms strong attractions to the ions, breaking up the ionic lattice. Covalent molecules, such as iodine, do not dissolve in water but do dissolve in non-polar solvents, such as cyclohexane. You can easily demonstrate this difference in relative solubility by adding a few crystals of iodine to a beaker filled with equal quantities of water and cyclohexane.
At 16–18 students look at the solute–solvent interactions in more detail and use them to explain trends in solubility in water of certain organic homologous series. Alcohol and carboxylic acid functional groups are polar and able to form hydrogen bonds with water. Short-chain molecules are therefore soluble in water because the hydrogen bonding dominates. In long-chain molecules the non-polar hydrocarbon chain dominates and the molecules are insoluble in water.
Older students also look at the thermodynamics of dissolving ionic salts in water. By comparing the energy needed to break up the ionic lattice with the energy released when individual gaseous ions are solvated, they can calculate the overall enthalpy of solution. They may also study solutions as examples of dynamic equilibria and investigate the effect of changes, like temperature and the addition of common ions, on the equilibrium.

Common misconceptions
Unfortunately, some typical investigations on the topic of solubility at 11–14 can lead to misconceptions at 14–16. Powdered sugar may dissolve faster than a sugar cube, but the solubility of the sugar isn’t affected. Revisit these investigations with older students to address any misconceptions.
Once dissolved, students may think the solute has disappeared because it can no longer be seen. You can overcome this misconception by encouraging students to relate the learning to what they have experienced in everyday life. If you swim in the sea and then dry out you can feel the solid salt on your skin. It was always in the sea; just dissolved. Revisit this learning when looking at separating mixtures by evaporation and the quantitative analysis of samples of saltwater.
Suggestions for your teaching
When first learning about concentration, it is easy for students to rely on inputting numbers into an equation without having a good understanding of what those numbers mean. You can avoid this by introducing the concept of concentration by using a series of simplified particle diagrams.

Manage cognitive load by starting with amount in particles and units for volume that students are familiar with prior to introducing mass, moles and decimetres cubed. Draw each solution live and change the amount of solute, volume of solvent or concentration of the final solution incrementally, to help students develop a better understanding of how to use and manipulate the equation when you finally introduce it.
Students need to understand how temperature affects solubility when separating a soluble salt from a solution by crystallisation. The time constraints of a typical lesson mean that students often leave an evaporating dish with their saturated solution of copper sulfate one day and return to it the next lesson to find dry crystals. As a result, they fail to recognise the importance of cooling on the process.
Introduce solubility curves at this point and structure questions to support students to move between numerical information about solubility and what they can observe. For example, show students a solution of ammonium chloride with a concentration of 50 g in 100 cm3 of water being cooled from 80 to 20°C. Crystals form. Ask them why. Although an in-depth understanding of crystallisation is not needed at 14–16, a good understanding of how temperature affects solubility is essential if students are to master the art of recrystallisation at 16–18 and in further studies.
Resources for your classroom
- Identify learning gaps and misconceptions with these worksheets on solubility.
- Discover what your learners know already with the diagnostic questions and response activities in Key concept 1: comparing solubility from the BEST resource on substances and properties.
- Model what happens when a solution becomes saturated on a particle level with this PhET simulation.
- Use this solubility experiment with your learners and get them to create their own solubility curve for ammonium chloride.
- Identify learning gaps and misconceptions with these worksheets on solubility: rsc.li/4c3XHFY
- Discover what your learners know already with the diagnostic questions and response activities in Key Concept I from the BEST resource on substances and properties: bit.ly/4hFp1eH
- Model what happens when a solution becomes saturated on a particle level with this PhET simulation: bit.ly/4j1zKRX
- Use this solubility experiment with your learners and get them to create their own solubility curve for ammonium chloride: rsc.li/41QEkLK













1 Reader's comment