Discover ideas for enhancing practical work on separating mixtures by filtration, evaporation and crystallisation
David Paterson updated this article on 17 April 2026.
The physical processes of separating mixtures by filtration, evaporation and crystallisation are key to many industrial and research activities. For example, water purification, making edible salts and pharmaceutical formulations all require aspects of these techniques. When research chemists are making new substances, they will often use purification techniques as part of the synthetic process to allow them to carry out structural analysis.

Underlying principles
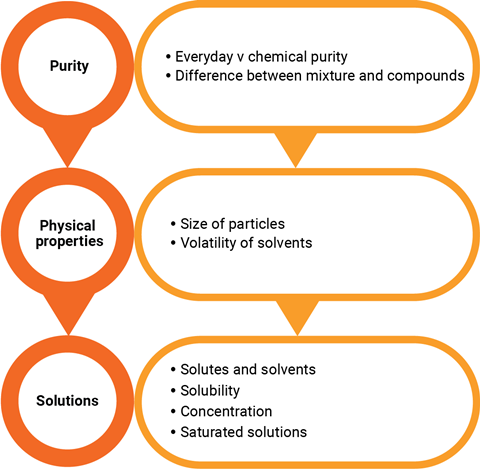
Separating substances is based on the principles of purity, physical properties and solutions (see flow chart). For example, chromatography (rsc.li/3Qtorsx) is a separation process based on solubility and distillation (rsc.li/4cPK2UH) is based on boiling point. This article looks at separation by particle size (filtration) and removal of volatile solvents (evaporation and crystallisation).
The concepts of purity and the difference between mixtures and compounds underlie all separation techniques. Discuss purity with your students using everyday drinks as a context (rsc.li/4my2uVf). Ask them to analyse the labels of bottled water or juices that claim to be 100 per cent pure, yet show various substances are present. Among other components, mineral waters contain varying levels of dissolved salts, and orange juices contain sugars, protein and vitamin C.
Separating substances is based on the principles of purity, physical properties and solutions (see flow chart below). For example, chromatography is a separation process based on solubility and distillation is based on boiling point. This article looks at separation by particle size (filtration) and removal of volatile solvents (evaporation and crystallisation).
The concepts of purity and the difference between mixtures and compounds underlie all separation techniques. Discuss purity with your students using everyday drinks as a context. Ask them to analyse the labels of bottled water or juices that claim to be 100 per cent pure, yet show various substances are present. Among other components, mineral waters contain varying levels of dissolved salts, and orange juices contain sugars, protein and vitamin C.
Use simple practical activities and demonstrations to help your students distinguish mixtures from compounds. Students can carry out the classic iron sulfide synthesis reaction, with access to CLEAPSS guidance for those in England, Wales and Northern Ireland (bit.ly/4mB7s3n) and SSERC guidance for its members in Scotland (bit.ly/4tWvY1D). Teachers can also demonstrate the reaction of aluminium with iodine (rsc.li/4tX2XTy). Students could repeat the simple separations of mixtures they have carried out at primary or 11–14 level. For example, separating sand, salt and sawdust (rsc.li/4eadDtp) or iron from breakfast cereal (rsc.li/4thUxG7).
You can easily demonstrate the importance of particle sizes by sieving a mixture of rice and caster sugar, and show the volatility of substances by using a few drops of propanone on a watch glass in different environments (rsc.li/4ch3Cck).
Use the context of the formation of saturated solutions of ammonium chloride to discuss the concepts of solution and saturation (rsc.li/484b8VB). Try this activity both qualitatively and quantitatively, introducing concentration and solubility calculations in later years. The Chemical misconceptions resources cover the differences between elements, compounds and mixtures (rsc.li/3OaJ5gt).
A relevant context to engage students – and link to social and economic aspects of chemistry – is the separation of metals’ ores. Rocks are mixtures of different minerals and must be separated to concentrate, and eventually purify, the desired metal. Physical processes such as crushing, sifting and flotation separate the components (bit.ly/4sAMMtW). The recycling industry uses similar processes to separate metals, glasses and plastics in household waste and when recycling cars.
Use simple practical activities and demonstrations to help your students distinguish mixtures from compounds. The classic iron sulfide synthesis reaction can be carried out by students, and the reaction of aluminium with iodine can be demonstrated. Students could repeat the simple separations of mixtures they have carried out at primary or 11–14 level. For example, separating sand, salt and sawdust or iron from breakfast cereal.
You can easily demonstrate the importance of particle sizes by sieving a mixture of rice and caster sugar, and show the volatility of substances by using a few drops of propanone on a watch glass in different environments.

Use the context of the formation of saturated solutions of ammonium chloride to discuss the concepts of solution and saturation. Try this activity both qualitatively and quantitatively, introducing concentration and solubility calculations in later years. The Chemical misconceptions resources cover the differences between elements, compounds and mixtures.
A relevant context to engage students – and link to social and economic aspects of chemistry – is the separation of metals’ ores. Rocks are mixtures of different minerals and must be separated to concentrate, and eventually purify, the desired metal. Physical processes such as crushing, sifting and flotation separate the components. The recycling industry uses similar processes to separate metals, glasses and plastics in household waste and when recycling cars.
Progression of ideas and skills
As students progress through school, their conceptual and procedural knowledge and skills are expected to develop.
| Secondary range | Types of learning areas |
|---|---|
|
Pre secondary |
Separation with sieves Formation of limescale (dissolved solids) Rock cycle – formation of sedimentary rocks Water cycle – filtering of water through rocks |
|
Early (approx. 11–14) and mid (approx. 14–16) |
Separation techniques for mixtures of substances including filtering and crystallisation The structure of matter and state changes, including evaporation |
|
Late (approx. 16–19) |
Filtration, including use of fluted filter paper Filtration under reduced pressure Recrystallisation |
Evaporation
Evaporation requires heat (or air movement above the sample) to drive off a volatile solvent. If the substance is a solid mixed in a solvent, begin by filtering (or decanting). If the substance is dissolved in a solvent, use crystallisation.
There are various techniques for heating a substance. For example, direct heating with a Bunsen burner or a sand bath, or by placing an evaporating basin over a beaker of heated water. Students should discuss and evaluate the different methods. For example, direct heating is quicker, but can superheat the solution, potentially degrading the solute and leading to splitting of the solid. Make links to the difference between distillation via direct heating and steam distillation, and the use of the latter in separating fragile components (rsc.li/41GaDgL).
Students could annotate diagrams of the different methods to consolidate their understanding, helping them learn the names and diagrams of the various laboratory apparatus.
There are various techniques for heating a substance. For example, direct heating with a Bunsen burner or a sand bath, or by placing an evaporating basin over a beaker of heated water. Students should discuss and evaluate the different methods. For example, direct heating is quicker, but can superheat the solution, potentially degrading the solute and leading to splitting of the solid. Make links to the difference between distillation via direct heating and steam distillation, and the use of the latter in separating fragile components.
Annotating diagrams of the different methods can help to consolidate students’ understanding, helping them learn the names and diagrams of the various laboratory apparatus (MS Word or pdf), while you can use a label-free worksheet in your classroom to help consolidate further (MS Word or pdf). You might also find the poster useful for providing a ready reference and further resources.
Have a set of questions ready to help focus your students’ thinking on the purpose and practicalities of the techniques.
| Technique | Possible questions |
|---|---|
|
Evaporation |
What observations tell you the evaporation is complete? What is the best heat source for this mixture? How does this evaporation fit into a larger separation procedure (eg filtration/crystallisation)? |
|
Crystallisation |
Where and why do crystals first form in the solution? What factors affect the size and quality of the crystals formed? Have solvent molecules formed part of the crystal structure? |
|
Filtration |
Why is the solid residue retained on the filter paper? How successful has the filtration been (how clear is the filtrate)? What is the advantage of a fluted filter paper? Is the residue fully purified once all the filtrate has passed through the filter paper? |
Crystallisation
Crystallisation occurs when the solvent of the solution evaporates and the concentration of the solute reaches saturation point. At this stage, the solute begins to precipitate out of solution. Under the right conditions, generally slow evaporation and a clear solution, the solute will crystallise. Demonstrate the principles of crystallisation with a saturated solution of sodium acetate. You can also use this activity to model the freezing process, and it can help to tackle misconceptions around the exothermic nature of freezing. A similar activity suitable for a student practical is freezing super-cooled sodium thiosulfate.
Recrystallisation is a key practical skill for post-16 students. You could show them a video to familiarise them with the process. 2-hydroxybenzoic acid has a low solubility at room temperature (2.5 g dm-3) and high solubility at 100°C (77.8 g dm-3), so it’s useful for demonstrating and practising this technique. Put recrystallisation in the wider context of organic synthesis by synthesising and purifying aspirin or paracetamol.
Crystallisation could form a project either for extension within class or perhaps as an activity for a science club. The RSC ran a global experiment in 2014 looking at the art of crystallisation. The project suggests various salts, including potassium nitrate and alum, and the resources include a useful crystal shape classification.
Crystallisation occurs when the solvent of the solution evaporates and the concentration of the solute reaches saturation point. At this stage, the solute begins to precipitate out of solution. Under the right conditions, generally slow evaporation and a clear solution, the solute will crystallise. Demonstrate the principles of crystallisation with a saturated solution of sodium acetate (rsc.li/4sF7KrB). You can also use this activity to model the freezing process, and it can help to tackle misconceptions around the exothermic nature of freezing. A similar activity suitable for a student practical is freezing super-cooled sodium thiosulfate (rsc.li/4cxrjfu).
Recrystallisation is a key practical skill for post-16 students (rsc.li/4mASgDt). 2-hydroxybenzoic acid has a low solubility at room temperature (2.5 g dm-3) and high solubility at 100°C (77.8 g dm-3), so it’s useful for demonstrating and practising this technique (rsc.li/4ewqkP0). Put recrystallisation in the wider context of organic synthesis by synthesising and purifying aspirin (rsc.li/4tUWU1D) or paracetamol (rsc.li/3QLK8nG).
Crystallisation could form a project either for extension within class or perhaps as an activity for a science club. The RSC ran a global experiment in 2014 looking at the art of crystallisation (rsc.li/3QeI5Zk). The project suggests various salts, including potassium nitrate and alum, and the resources include a useful crystal shape classification.
Want more resources for changes of state?
Explore our resources package, with everything you need to teach this topic at 11–14.
Practical problems and suggested solutions
Filtration requires various pieces of glassware that can cause confusion about what needs placing where, as well as what is poured where and when. Demonstrate the techniques and explicitly name the apparatus to reinforce this important knowledge. Despite being used commonly throughout their science education, filtration can cause students several problems.
Slow filtering of mixtures is generally caused by the filter paper becoming clogged with the residue. Advise students to allow their mixture to settle first, then decant the liquid (the supernatant) carefully into the filtering equipment. They could also filter with a coarser filter paper first, then a finer one as a serial filtration. Show them how to fold the filter papers in the fluted fashion, usually only taught at post-16, as this increases the surface area of filtration.
If the students are filtering under reduced pressure, show them how to check the seal formed between the funnel and the flask, and check that they have pre-wetted the filter paper before pouring in the mixture. Check also that the pump is drawing a good vacuum. If they are using a water pump, turn the tap on full to get the best pumping action possible. Mechanical pumps are superior but not commonly found in secondary chemistry labs.
Bring techniques together in context
The production of sweets is a good example. Aqueous sugar solutions at different concentrations have different properties. As a sugar solution is boiled, the water evaporates and the concentration of the sugar increases. As the boiling point is dependent on sugar concentration, the temperature of the solution increases steadily as concentration increases, from the soft ball (fudge) concentration (85 per cent) to hard crack (toffee, 99 per cent).
A more complex context, but one that can lead to many interesting discussions, is chocolate. Multiple crystal forms exist, providing many extension opportunities to stretch and challenge keen students. The overlap of chemistry, cooking and business makes studying chocolate a potentially interesting cross-department project.
The production of sweets is a good example (rsc.li/4cqV5Cu). Aqueous sugar solutions at different concentrations have different properties. As a sugar solution is boiled, the water evaporates and the concentration of the sugar increases. As the boiling point is dependent on sugar concentration, the temperature of the solution increases steadily as concentration increases, from the soft ball (fudge) concentration (85 per cent) to hard crack (toffee, 99 per cent).
A more complex context, but one that can lead to many interesting discussions, is chocolate (rsc.li/4cuPWJw). Multiple crystal forms exist, providing many extension opportunities to stretch and challenge keen students (rsc.li/4vA4nVm). The overlap of chemistry, cooking and business makes studying chocolate a potentially interesting cross-department project.
Drawing techniques together in one activity

Making copper sulfate crystals from copper oxide and sulfuric acid requires filtration, evaporation and crystallisation techniques – teachers in England, Wales and Northern Ireland can use CLEAPSS guidance, while those in Scotland who are members can use SSERC’s guidance. This practical can be carried out at various scales. The traditional method, involving heating sulfuric acid directly, has mostly been replaced by warming slightly more concentrated acid in a hot water bath, providing a lower risk method.
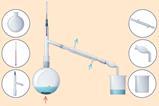
An alternative microscale method is also available. The reaction mixture is heated using a sand bath, both to increase the rate of the reaction and to evaporate some of the solvent from the copper sulfate solution. Next, the sample is filtred thorugh mineral wool in a syringe. Aside from the speed advantages, students can work individually, giving them time to focus on improving their manual dexterity. It is possible for students to produce cyrstals within a normal teaching lesson, as shown in the image above.
Making copper sulfate crystals from copper oxide and sulfuric acid requires filtration, evaporation and crystallisation techniques. There are various scales at which this practical can be carried out, with CLEAPSS guidance for England, Wales and Northern Ireland (bit.ly/4tbcd6m) and SSERC for members in Scotland (bit.ly/4tWOOFX). The traditional method, involving heating sulfuric acid directly, has mostly been replaced by warming slightly more concentrated acid in a hot water bath, providing a lower risk method (rsc.li/3OMg8rj).
An alternative microscale method is also available (rsc.li/4cQ1oRn). The reaction mixture is heated using a sand bath, both to increase the rate of the reaction and to evaporate some of the solvent from the copper sulfate solution. Next, the sample is filtred thorugh mineral wool in a syringe. Aside from the speed advantages, students can work individually, giving them time to focus on improving their manual dexterity. It is possible for students to produce cyrstals within a normal teaching lesson, as shown in the comparison of crystallisation image.
Downloads
Apparatus diagrams - with labels
Word, Size 0.84 mbApparatus diagrams - with labels
PDF, Size 0.39 mbApparatus diagrams - without labels
Word, Size 0.82 mbApparatus diagrams - without labels
PDF, Size 0.1 mb

Teaching practical science

Boost your confidence and your learners’ outcomes with these articles focused on practical lessons
- 1
- 2
- 3
- 4
- 5
- 6
 Currently
reading
Currently
reading
How to teach evaporation, filtration and crystallisation


























No comments yet