Use this to help your students make all the connections they need to ace covalent bonding
A chemical bond holds the parts of a chemical structure together. When the bonding is between two non-metallic atoms, a covalent bond is formed.
Covalently bonded species can fall into one of two categories:
- elements, where all the atoms are of the same non-metal
- compounds, where two or more different non-metallic atoms are bonded together.
Different models are used at 14–16 to represent covalently bonded species (see Figure 1 for methane).
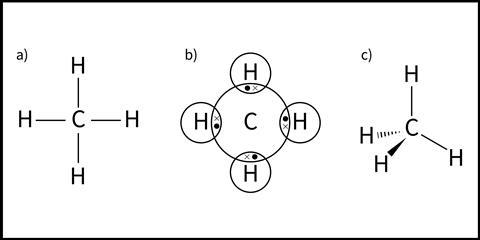
In the simplest model (Figure 1a) a covalent bond is represented by a straight line between the atoms shown by their element symbols. In dot and cross diagrams, commonly introduced at 14–16 (Figure 1b), each atom’s outer shell electrons are shown in circular orbits. Covalent bonds are represented by a shared pair of electrons in an area of overlap.
Neither of these models show the 3D shape of the molecule. Jumping between the 2D representation to the 3D shape of a molecular model can be conceptually challenging for students. A third model (Figure 1c) goes some way to overcome this difficulty. Dashes and wedges represent bonds going into and coming out of the page respectively.
Neither of these models show the 3D shape of the molecule. Jumping between the 2D representation to the 3D shape of a molecular model can be conceptually challenging for students. A third model (Figure 1c) goes some way to overcome this difficulty. Dashes and wedges represent bonds going into and coming out of the page respectively.
What you need to know
At 14–16, students will typically describe a covalent bond as a shared pair of electrons. They may go on to explain that the positively charged nuclei of the bonded atoms are attracted to the shared pair of electrons by electrostatic forces, which makes covalent bonds very strong. But what about the repulsion between the electrons?
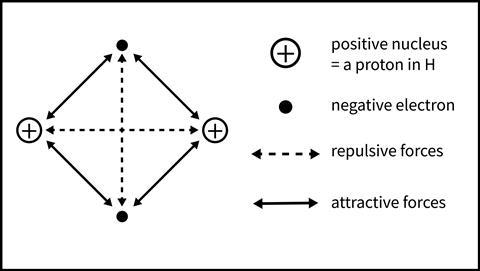
Students will understand the bonding in a simple covalent molecule more clearly by considering all the electrostatic interactions involved. Figure 2 shows the interactions between the two nuclei and electrons in the diatomic molecule, H2. Within the molecule there are both attractive (proton–electron) and repulsive (electron–electron and proton–proton) forces. The strength of these forces varies with the inverse of the square of the distance between the particles involved. Therefore, as the atoms approach each other, the strength of the attractive and repulsive forces change. At a certain distance, the equilibrium internuclear distance, the net attractive interaction is maximised. With the atoms this distance apart, the molecule is most stable.
Students will understand the bonding in a simple covalent molecule more clearly by considering all the electrostatic interactions involve, such as the interactions between the two nuclei and electrons in the diatomic molecule, H2. Within the molecule there are both attractive (proton–electron) and repulsive (electron–electron and proton–proton) forces. The strength of these forces varies with the inverse of the square of the distance between the particles involved. Therefore, as the atoms approach each other, the strength of the attractive and repulsive forces change. At a certain distance, the equilibrium internuclear distance, the net attractive interaction is maximised. With the atoms this distance apart, the molecule is most stable.
Resources for your classroom
See all of our supporting resources for covalent bonding at a glance in our structure and bonding resources package for learners aged 14–16.
- Help your 14–16 students master simple dot and cross diagrams with covalent bonding tiles.
- Identify misconceptions in student understanding of covalent bonding at 11–14 with this BEST resource on particles and structure.
- Use this activity looking at energetic stability to develop students’ understanding of covalent bonding.
Resources for your classroom
- Help your 14–16 students master simple dot and cross diagrams with covalent bonding tiles: rsc.li/3TwEVfU
- Identify misconceptions in student understanding of covalent bonding at 11–14 with this BEST resource on particles and structure: bit.ly/3VFGsCg
- Use this activity looking at energetic stability to develop students’ understanding of covalent bonding: rsc.li/3yWrjT9
The model described above is limited because it represents the protons and electrons as point charges. To better understand the covalent bond, it’s necessary to look at a more sophisticated model for atomic structure. At post-16, electrons are no longer considered to be a point particle, rather a cloud of negative charge. An electron fills a volume in space, called its atomic orbital. Density plots indicate the probability of finding an electron in a given space. The orbital diagrams familiar at post-16 are a simplification of density plots. A single line is drawn to represent the outer edge of an area where there is a 90% chance of finding the electron. The shapes of orbitals and how they overlap dictate the strength and type of covalent bonds formed.
Clearing up misconceptions
Dot and cross diagrams and the idea of ‘sharing’ electrons can make students think electrons ‘belong’ to a specific atom. Students can see shared pairs of electrons as ‘glue’ that ‘joins’ atoms together. Simple molecular modelling kits can embed this misconception – covalent bonds are represented as physical objects holding spherical atoms together.
What the model lacks
Ideas that can’t be explained using the 14–16 model of covalent bonding include:
- the difference between a single and double covalent bond
- the reactivity of alkenes
- delocalisation of electrons in conjugated systems
- dipole moments and polar bonds.
It’s important to emphasise the electrostatic nature of the attractions that hold the two atoms together in a covalent bond. From relatively early on in their secondary education, students are familiar with the idea of opposite charges attracting and like charges repelling. Once students have met dot and cross diagrams, I start the next lesson with this activity, taken from Covalent bonding teaching resources. I ask them to annotate the diagram of a hydrogen molecule (Figure 3) to show all the attractive and repulsive forces that would exist. This simple activity encourages students to think beyond the shared electrons idea for covalent bonding.
It’s important to emphasise the electrostatic nature of the attractions that hold the two atoms together in a covalent bond. From relatively early on in their secondary education, students are familiar with the idea of opposite charges attracting and like charges repelling. Once students have met dot and cross diagrams, I start the next lesson with an activity on covalent bonding (bit.ly/3TBboBs). I ask them to annotate the diagram of a hydrogen molecule (Figure 3) to show all the attractive and repulsive forces that would exist. This simple activity encourages students to think beyond the shared electrons idea for covalent bonding.
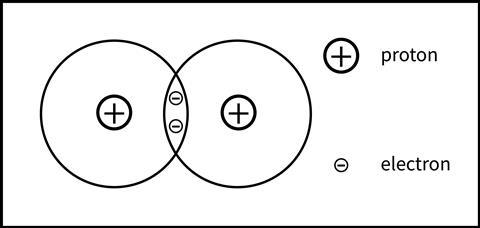
The idea that substances are held together by electrostatic forces of attraction is described as one of 13 powerful ideas – the ideas being the most conceptually important parts of the science we want students to acquire at school. This shift in focusing on electrostatics can be applied to all types of bonding at 14–16. Students will have deeper understanding of the connections between different types of bonding, and progress to more complex ideas about bonding encountered at post-16.
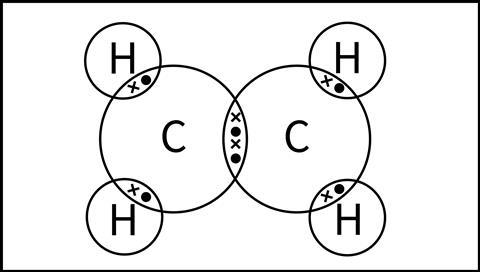
At this stage, we would draw a molecule of ethene as a dot and cross diagram, as shown in Figure 4. Although this model shows the connectivity and explains the existence of a double bond between the two carbon atoms, it doesn’t explain the molecule’s shape or its reactivity.
The idea that substances are held together by electrostatic forces of attraction is important, and this shift in focusing on electrostatics can be applied to all types of bonding at 14–16. Students will have deeper understanding of the connections between different types of bonding, and progress to more complex ideas about bonding encountered at post-16.
At this stage, we would draw a molecule of ethene as a dot and cross diagram. Although this model shows the connectivity and explains the existence of a double bond between the two carbon atoms, it doesn’t explain the molecule’s shape or reactivity.
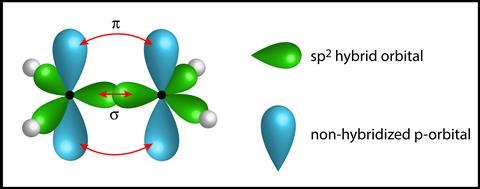
To fully understand the distribution of the electrons in ethene, students need to understand the process of hybridisation, which is significantly beyond the 14–16 curriculum. However, the concept of orbital overlap can be introduced. Each carbon atom has four electrons to share. Three are held within identical orbitals (sp2) which orientate as far apart as possible to minimise electrostatic repulsion, and the fourth is held within a p orbital. A simple diagrammatic representation (such as Figure 5) allows the students to see the shape of the ethene molecule and understand its reactivity owing to the high energy electrons held within the weaker pi bond formed from the ‘sideways’ overlap of two parallel p orbitals.
To fully understand the distribution of the electrons in ethene, students need to understand the process of hybridisation, which is significantly beyond the 14–16 curriculum. However, the concept of orbital overlap can be introduced. Each carbon atom has four electrons to share. Three are held within identical orbitals (sp2) which orientate as far apart as possible to minimise electrostatic repulsion, and the fourth is held within a p orbital.
Take-home points
- When teaching bonding, shift the focus away from atoms needing to gain a full outer shell; rather focus on the electrostatic nature of chemical bonding.
- Consider teaching metallic and ionic bonding first (as the electrostatic interactions are more obvious), before moving on to covalent bonding.
- Encourage students to think about electrons existing in clouds around a molecule by showing students space-filled models for molecules, as well as the simpler ball-and-stick models. Try using MolView.
- As you introduce the different models for covalent bonding, be explicit about their strengths and weaknesses.
- Encourage interested students to read further about the Lennard Jones potential for modelling interactions between particles.
Take-home points
- When teaching bonding, shift the focus away from atoms needing to gain a full outer shell; rather focus on the electrostatic nature of chemical bonding.
- Consider teaching metallic and ionic bonding first (as the electrostatic interactions are more obvious), before moving on to covalent bonding.
- Encourage students to think about electrons existing in clouds around a molecule by showing students space-filled models for molecules, as well as the simpler ball-and-stick models.
- As you introduce the different models for covalent bonding, be explicit about their strengths and weaknesses.
- Encourage interested students to read further about the Lennard Jones potential for modelling interactions between particles: bit.ly/3VCWdcV














No comments yet