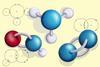
Help learners to visualise the shape and structure of simple covalent molecules with these manipulative covalent bonding tiles
Once learners have mastered the skill of drawing electron configuration diagrams, they are ready to move on to bonding diagrams using the dot-and-cross method. This resource helps learners to visualise the position of atoms within simple molecules by experimenting with layouts and moving the tiles to complete the covalent bonds.
This resource accompanies the Education in Chemistry infographic How to draw dot and cross diagrams.
The four tasks outlined in this resource are examples of how the tiles could be used to complement topics at various points in the chemistry course for 14-16 year old learners. It is not expected that these would be delivered as part of a single lesson or sequence. The tiles may also be useful at other points in the course that are not outlined here.

Instructions for teachers
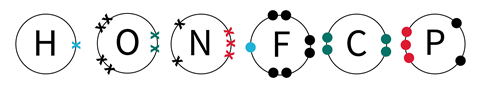
This resource contains a sheet of printable covalent bonding tiles which can be printed and cut out into individual tiles to support learners when introducing covalent bonding. The electrons are identified using colour coded dots and crosses to help learners identify single, double and triple bonds. To avoid misconceptions learners will need to be reminded that, although different symbols are used, they all represent a single electron in the outer shell of the atom.
Task 1: Building bonds
In this task, learners familiarise themselves with the tiles in the resource and practice arranging them to make simple covalent molecules. This task is deliberately open-ended and there are a large number of possible arrangements for each of the steps in the task. Learners will get used to finding the correct tiles to complete each bond and the arrangements of the tiles which they may sometimes need to place at an angle.
Task 2: Simple covalent molecules
In this task, learners are asked to build named molecules based on their chemical formula. In the case of molecules containing oxygen, carbon and nitrogen, learners will need to choose the correct tile to show single, double or triple bonds as required.
Task 3: Hydrocarbons
In this task, learners use the tiles to explore patterns in the homologous series of alkanes and alkenes. Learners should already be familiar with using the resource (the different stages in task 1 help to explore this) before applying the tiles to chemistry concepts. The tiles can be used to demonstrate the general formulas for the alkanes and alkenes.
Task 4: Polymerisation
In this final task, learners are asked to create a stop motion animation or GIF to illustrate the process of polymerisation. They will need to use creativity and secure knowledge of structure and bonding to produce their animation.

Instructions for learners
Position the tiles to arrange the atoms into molecules. The electrons are colour coded to help identify the type of bond and where the bond will be. The blue dots and crosses will form a single covalent bond, the green dots and crosses will form a double covalent bond and the red dots and crosses will form a triple covalent bond. The black dots and crosses are not involved in bonding.
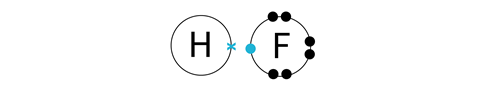
Match the colours on the dot and cross tiles to complete the bond.
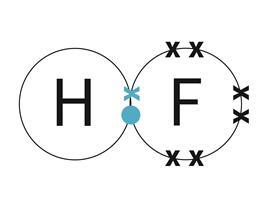
Once you are happy with the arrangement of your atoms within your molecule, draw the dot and cross diagram.
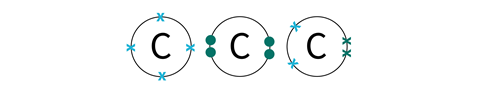
Some of the atoms will have different arrangements depending on the type of bonds that they will form and the shape of the resulting structures. You will need to choose carefully and try different combinations to find the best fit for your molecule.
Match, move and explore different combinations.
Downloads
Covalent bonding tiles teacher notes
Editable handout | Word, Size 2.19 mbCovalent bonding tiles teacher notes
Handout | PDF, Size 3.43 mbCovalent bonding tiles student sheet
Editable handout | Word, Size 2.56 mbCovalent bonding tiles student sheet
Handout | PDF, Size 0.9 mb

































3 readers' comments