Jump on the escape room bandwagon, and challenge your students with these puzzles

In a typical escape room, participants are locked in a room and must solve puzzles as a team to escape. While this is not practical in schools, in your classroom you can challenge students as a group to solve various chemistry-based puzzles to open a locked box containing a reward, allowing them to develop problem-solving, knowledge and practical skills in a novel way. Here are some examples – hopefully they’ll inspire you to develop your own puzzles with your students.
Materials
A small tool box with a reward inside, a hasp lock and six assorted number or word locks per group are required. The hasp lock can hold another six locks that have to be unlocked to open the box.
Puzzle 1 – Jigsaw

In this activity, students use their knowledge of chemistry to complete a card-matching jigsaw (download this example below), and then use a UV torch to reveal the code which has been written on the pieces with a UV marker pen. Make sure that the numbers are on a few different pieces as students will try shortcuts like discarding pieces that don’t fluoresce in UV light. As part of a recap, students can use the UV torch to examine banknotes and see how UV light is used to detect and prevent forgeries.
Puzzle 2 – Periodic table guess who

In this activity, the faces on a Guess Who game have been changed to element symbols using sticky labels (download the worksheet below). The students eliminate the elements according to a list of clues given on a sheet of paper. The elements must be rearranged as letters to spell a country, in this case China. The word China can be then be used as a password to open a word lock.
Puzzle 3 – Thermochromic revelation

Students are provided with a sheet of paper on which a three-digit code has been painted in red paint. Other numbers have been painted with red thermochromic paint. When the paper is heated, the thermochromic paint will change to white and reveal the code. As a recap, this can be used to introduce smart materials and the use of thermochromic paint as a temperature sensor.
Puzzle 4 – Microscale chemical changes
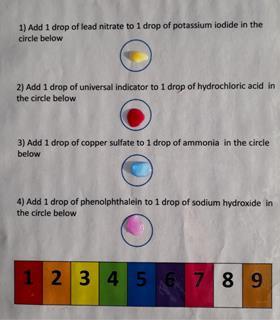
In this activity, students add drops of two different chemicals to each other using the instructions (download the worksheet below) on a laminated sheet and note the colour. The colour will correspond to a number. This will give the code required to unlock the box.
For younger pupils unfamiliar with the names of chemicals, this is a good literacy and practical exercise as they need to read the names carefully, follow instructions and observe. For older pupils the concepts of precipitation, neutralisation and transition complex formation can be introduced or reinforced.
Puzzle 5 – UV revelation
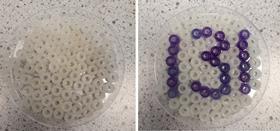
For this puzzle, students are given a petri dish which contains UV colour changing beads that have been arranged into a three-digit number and stuck on the bottom using PVA adhesive. Surrounding the UV beads are white pony beads.
If you can’t get white pony beads, use the UV colour changing beads that turn the lightest colour instead and use the code darkest for the code; see the purple image.
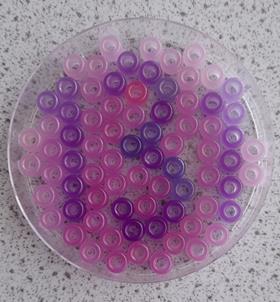
Give your students a clue such as ‘Give the beads a dose of UV, then the code is clear to see’.
The pupils should link UV with sunlight and take the beads outside. The UV beads will change colour and reveal the code. This can lead to a discussion of the effects of UV light and the chemistry of sun cream.
Puzzle 6 – Conductivity code

Give students nine materials with an assigned number; they must find out which ones conduct electricity, to get the code. The inclusion of electrolycra and carbon fibre can be used be trigger discussion about wearable technology as well as the chemistry of what makes a substance conduct electricity. Download the worksheet below.
Taking it further
The difficulty of the puzzles can be tailored to the age and stage of the pupils. For older pupils the code could be numbers missing from a balanced equation or the answer to a chemical calculation.
This article was updated, 18 August 2023.
Want more ideas and resources? Take a look at our follow-up article: Escape the classroom – again!
Downloads
Escape the classroom worksheets
Word, Size 0.38 mbEscape the classroom worksheets
PDF, Size 0.37 mb
Escape the classroom

Be inspired to design an escape room experience for students with these chemistry related puzzles
- 1
- 2
- 3
 Currently
reading
Currently
reading
Basic puzzles
- 5
- 6
- 7






















7 readers' comments