Dorothy Warren describes some of the difficulties with teaching this topic and shows how you can help your students to master aspects of quantitative chemistry
Many teachers find quantitative analysis (ie titrations) a tough topic to teach. This may be due to the scary aspects of managing the practical work or the teaching of titration calculations, which requires a good understanding of the mole; another tough topic for both students and teachers.
A titration is simply a technique used by analytical chemists to determine the concentration of an unknown solution by reacting it with a solution of accurately known concentration. Put like that, it sounds really easy and straightforward, so why does it cause so many teachers anxiety? Students often experience a number of difficulties when they are first introduced to titrations, from handling the equipment correctly to the ability to follow instructions (fig 1)! Some schools lack enough good quality equipment, forcing students to work in large groups. Others battle with leaky burettes, leading to meaningless results full of errors. It is easy to see how progress can be hindered.
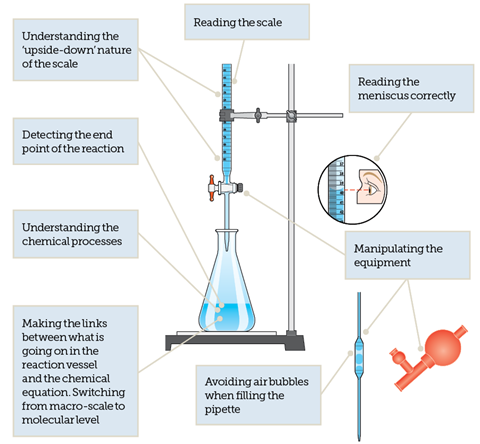
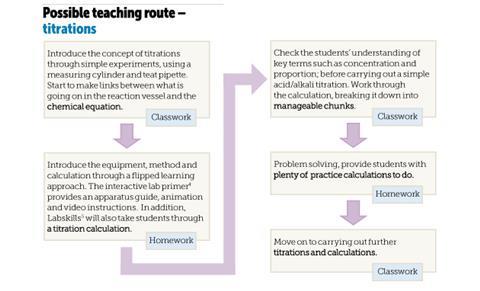
In order to overcome some of these difficulties the topic needs to be broken down into manageable chunks. It is really important that as teachers we gradually develop the students’ skills and confidence with each of the component aspects of titrations by providing appropriate scaffolding. Being able to handle full-scale titrations in the classroom combined with the necessary calculation work requires careful planning and sequencing of ideas (see Possible teaching route – titrations).
Not enough specialist equipment?
If lack of good quality specialist equipment, burettes and pipettes is an issue in your school, you could start to introduce the concept of titrations by simply using dropper pipettes, measuring cylinders and small beakers. This will also provide some simple data that could be used to introduce the calculations. Next, try moving on to micro burettes. They are very easy to make from syringes, graduated pipettes and silicone tubing (fig 2). The micro burette can be used in conjunction with a 1 or 2 cm3 syringe (instead of a pipette) to carry out a titration. Micro titrations give surprisingly good results. If you have not already tried out the technique do have a go. Not only will all the class be able to do the practical but you will also save on your chemical costs! Further experimental details can be found out the practical chemistry website.1
Students can’t follow instructions
Teaching titrations is a good opportunity to put flipped learning into practice,2,3 since there are good quality video methods and simulations4 readily available. Before the actual lesson, direct your students to this material so they can learn how to carry out a titration. Your students should then arrive in the lesson aware of the practical equipment, method and calculations needed to do a titration. This will have the knock on effect of freeing up some teacher time during the lesson to support the development of practical skills through working with small groups and to challenge understanding through targeted questioning.
So what about the mole?
Many of the difficulties associated with the concept of the mole start with its definition. When taking a closer look this is hardly surprising as chemists themselves do not agree on how the mole should be defined. It has been a topic of debate within the chemical community for over 60 years. One of the difficulties lies within the fact that the mole has three meanings:
- an individual unit of mass;
- a portion of substance; and
- a number.
Definition of the mole
SI unit of amount of substance, equal to that quantity containing as many elementary units as there atoms in 0.012 kg of carbon-12
To try and make sense of this definition, teachers often try to adopt a simplistic approach by thinking of the mole as a ‘counting unit’, which of course does not fit any of the above meanings. In order to understand the mole, students need to be presented with a clear approach based on making connections between the amount of substance and a numerical quantity. Students need to appreciate that moles link the substances in a chemical equation to the amounts needed in practice. However, if the mole is taught purely as an abstract mathematical idea, and this chemical context is lost, this too can introduce a whole set of new difficulties, especially to the students who already struggle to manipulate numbers, symbols and formulae.
Key findings from educational research:5 Why students find quantitative chemistry difficult
- Chemists do not agree on how the mole should be defined.
- The mole is taught as an abstract mathematical idea.
- Students lack secure understanding of preliminary concepts including:
- Chemical reactions produce new substances
- Matter is made from tiny particles invisible to the naked eye
- Chemists need to be able to measure amounts of substances accurately in order to be able to control a reaction.
- Avogadro’s number cannot be‘seen’.
Teachers must take time to introduce the mole to their students, develop the ideas and finally embed the ideas over a period of time (see Possible teaching route – the mole).

Dorothy Warren is an independent science education consultant based in the UK. Dorothy would like to acknowledge and thank Kay Stephenson for her input into this article
More CPD
This topic is covered in one of the Royal Society of Chemistry’s Developing Expertise in Teaching courses, designed to support you throughout your teaching career. It is available as both an online course and a face to face workshop.
To find out more about the CPD for Teachers courses on offer, visit www.rsc.org/Education/CPD
References
- http://bit.ly/1rT1QnK
- Education in Chemistry, September 2013, p14 (Flipping lectures and inverting classrooms)
- Education in Chemistry, November 2013, p8 (Flipped teaching in practice)
- http://bit.ly/1I3jNom
- V Kind, Beyond appearances: Students’ misconceptions about basic chemical ideas(2nd ed.), p50–53. Royal Society of Chemistry, 2004
- Iron and sulfur reaction
- http://bit.ly/1pY4cjK








1 Reader's comment