An early example of how blue skies research by Pierre and Marie Curie led to the treatment of previously incurable cancers
- In the early 1900s radium was used to reach deep-seated cancers that x-rays couldn't reach
- Eventually better treatments were found, but radium was used up until the 1980s
One hundred years ago, in 1911, Marie Curie was awarded her second Nobel prize, this time for chemistry:
...in recognition of her services to the advancement of chemistry by the discovery of the elements radium and polonium, by the isolation of radium and the study of the nature and compounds of this remarkable element.1
Radium and its decay product, radon, were used in medicine for well over half a century until being supplanted by radioactive isotopes prepared in nuclear reactors. This account describes their early use.
Discovery
In 1895, physicist Wilhelm Roentgen was passing a high voltage through an evacuated glass tube as part of his research on cathode rays. He surrounded his tube with a light-tight layer of cardboard, but noticed that a screen coated with barium platinocyanide, some 2.5 metres away, was fluorescing. Cathode rays travel only about 10cm outside a vacuum, so these must be some new type of ray, which he called x-rays. He found these rays could:
- ionise air and discharge an electroscope,
- penetrate flesh and reveal internal structures, such as bones, and objects such as bullets,
- give rise to deep-seated burns.
This last finding was discovered by chance as the early researchers were unaware of the energy of x-rays. Repeated exposure damaged their tissues, sometimes causing cancer.
In January 1896, Chicago physician Emile Grubbe, having noted the destructive effect of the rays on his own skin, applied them in the hope of "burning out" a patient's breast cancer. Although this was unsuccessful, by November that year x-rays were being used to cure superficial cancers, such as skin cancers, which were more accessible to the radiation.
A month or two after Roentgen's discovery, Henri Becquerel was working on the phosphorescence of uranium salts. Becquerel had left some potassium uranyl sulfate in contact with photographic film that was sealed in black paper to protect it from light. On developing the film he was surprised to find the film had been 'fogged'. He wrote:
When one then develops the photographic plate, one recognizes that the silhouette of the phosphorescent substance appears in black on the negative. If one places between the phosphorescent substance and the paper a piece of money or a metal screen pierced with a cut-out design, one sees the image of these objects appear on the negative . One must conclude from these experiments that the phosphorescent substance in question emits rays which pass through the opaque paper and reduces silver salts.2
The effect, of course, was due to the uranium itself, rather than any phosphorescence. Nevertheless, it was the world's first report of radioactivity. There were similarities with Roentgen's x-rays, which could also blacken photographic film, and both could discharge an electroscope, indicating they caused ionisation of the air.


Marie Curie, working at the Sorbonne, Paris, decided that Becquerel's radioactivity might prove a fruitful field for investigation. She used an electrometer (an improvement on the electroscope, though both depend on the ability of the radiation to ionise air) to detect and quantify the radioactivity.
Her first success, in 1898, was finding that thorium, like uranium, was radioactive. In the same paper she reported that pitchblende, an ore consisting mainly of UO2, was three times as active as uranium oxide, U2O5.
Correctly supposing that some other, as yet unknown, species was responsible for this enhanced activity, Marie Curie spent the next four years isolating it from pitchblende. Marie and her husband Pierre named this mystery substance radium in 1898.
Success came in 1902, when they announced they had isolated 0.1g of pure radium chloride, RaCl2, from almost 3 tons of pitchblende. This was powerfully radioactive; Pierre Curie estimated that it was about a million times more radioactive than uranium.
Although 'radiographs' equivalent to x-ray photographs could be produced, exposure times remained discouragingly long because radium was then only available in milligram quantities. Moreover, the more highly energetic γ-rays from the salt penetrated bone better than x-rays, reducing the contrast between bone and flesh in the radiographs, additionally discouraging further development for medical use.
Even before the pure salt had been isolated, it was apparent that radium, like x-rays, could 'burn' skin and flesh. In 1900 Friedrich Walkoff reported:
Furthermore, radium owns astonishing physiologic properties. An exposure of the arm to two 20 minute sessions has produced an inflammation of the skin which has now already lasted two weeks, and exhibits the same aspect as obtained after a long exposure to x-rays.3
This result prompted both Henri Becquerel and Pierre Curie to carry out some confirmatory work which they announced the following year. (The fact that Becquerel received his burn from a test tube of radium-rich barium chloride being transported in his waistcoat pocket leads one to think that the experiment was serendipitous rather than premeditated.)
When describing this work in a public lecture at the Royal Institution in 1903, Pierre Curie suggested that radium's ability to induce deep flesh burns might have potential in cancer therapy. Indeed, work on medical applications of radium had already started, albeit initially on non-cancerous skin conditions.
Lupus vulgaris is a form of tuberculosis that gives rise to painful skin lesions, particularly on the face. Previously untreatable, in 1896 the Danish physician, Niels Finsen found it could be cured by applying high-powered ultraviolet light. It was but a short step to test the rays emanating from radium salts on this disease. Pierre Curie lent a sample of radium sulfate, sheathed in rubber, to Parisian physicians Henri Danlos and Paul Bloch to do this, and they reported their success in 1901.
Soon radium was being used, curatively, on a variety of skin cancers. Most susceptible were 'rodent ulcers' (basal cell carcinomas). Dr J. H. Sequeira recollected in 1915:
Small rodent ulcers, and especially those which are freely moveable, react remarkably well to radium. This agent has a decided advantage over x-rays in certain situations, because it can be more readily applied....in the neighbourhood of the eye or in the angle of the nose. As far back as 1902 I was using radium for rodent ulcers.... The introduction of plates upon which the radium salt is spread and then covered with varnish has enabled us to treat with greater ease a large number of these superficial malignant growths.4
Burning out the cancer
For deeper-seated cancers, inaccessible to x-rays, radium treatment had obvious advantages, as sources could be placed within body cavities.
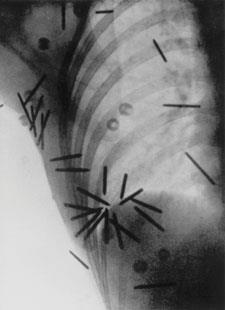
In 1904 John MacLeod, a physician at Charing Cross Hospital, was devising radium applicators for internal use. Fig 1 shows one designed to treat throat cancer. The lower end of a flexible metal tube contained a glass phial of the radium salt. The "bobbin" is made from rubber and the patient bites on this to position the radium accurately within the neck against the tumour. The applicator could also be used for uterine cancers:
...the cervix (had) a large mass of growth with a large crater-like cavity. Twelve exposures of half an hour each with a 'strong' tube...seemed to have a beneficial effect in breaking down and diminishing the malignant tissue.5
The prostate is a solid gland and is subject to cancer. From 1911 it was being treated by a thin catheter applicator, similar to MacLeod's, for radium insertion via the urethra. However from 1917 Benjamin Barringer was using hollow needles containing radium salts for direct, temporary, implantation:
While many excellent results have been obtained with radium in the treatment of superficial cancers, the results in the deeper seated neoplasms have not been very encouraging, owing largely to the difficulty of bringing the radium directly into contact with the involved areas. It is my belief that this is accomplished in cancer of the prostate by exposing the tumor both above and below and by inserting multiple needles, containing radium, in different directions, thus bringing the radium into contact with the most remote parts of the growth.6
One step up from this treatment was the use of radon gas, a decay product of radium. A 'radium cow' or sample of radium would continually yield a supply of the nuclide. With a half-life of only 3.8 days, radon "seeds" (Rn gas contained in thin walled gold tubing, crimped and cut into units up to 1cm in length) could be left permanently implanted in the body. Their use in prostate brachytherapy dates from 1926. The technique is still used today, but the radon has been replaced, typically, by the radionuclide 131Cs (produced in a nuclear reactor; half-life = 9.7 days).
Radium salts as medicines
The seemingly miraculous properties of radium and its compounds prompted investigations into other potential medical uses, including:
- hypertension (raised blood pressure) using doses of 10-50 mg, intravenously;
- pain associated with diabetic neuritis;
- arthritis;
- rheumatism and gout (by drinking spa-water, rich in radon);
- pain, in cases of inoperable cancer (subcutaneous injections of about 20 mg of RaSO4);
- tuberculosis of the lung, by breathing in the emanations (radon gas) from radium.
However, even by the late 1920s it seemed unlikely that any of these would be successful, as proved the case. Nonetheless some experimental work on relieving intractable pain by radium is currently underway.7
Unsurprisingly, such sober consideration did little to restrain the manufacturers of health-foods, patent medicines and surgical appliances, who sought to exploit the radioactive properties of radium (and thorium) for a quick financial reward.
Doramad's Radioactive Toothpaste (Fig2) claimed that radiation 'increases the defence of teeth and gums' and Revigators (ceramic jar-like water containers contained a minute source of radium continually giving off radon) were advertised "to create healthful radioactive water". Men were urged to take Vita Radium Suppositories, which purported to make "weak and discouraged men...bubble over with joyous vitality"; and if these did not have the desired effect, they could resort to the Scrotal Radiendocrinator. This consisted of sheets of blotting paper soaked in a dilute solution of radium salts, dried and sandwiched between plastic, and then held in position by credit-card sized rectangles of gold gauze. If you felt the need for enhanced sexual virility, you followed the manufacturer's advice 'to wear the adaptor like any other athletic support. This puts the instrument under the scrotum as it should be. Wear at night. Radiate as directed.'


In truth, the very low quantities of radium meant these products did little harm, apart from taking money off the gullible. More sinister was Radithor. This patent medicine came in 15cm3 bottles, each guaranteed to contain two microcuries of radium (one curie equates roughly to the activity of one gram of 226Ra and 1μm Ci = 3.7x104 disintegrations per second). Advertised as a 'cure for the living dead', a USA industrialist Eben Byers drank three bottles a day to relieve pain in his arm sustained by falling out of bed. Time Magazine reported in 1932:
The dope eased the arm pain, braced Byers up. He enthusiastically recommended it to friends, sent them cases of it, even gave some to one of his horses. Last week Eben Byers died in Manhattan of radium poisoning. Eighteen months ago, after hundreds of drinks of the radium tonic, he began having pains in his jaw, severe headaches. Dr. Joseph Manning Steiner, Manhattan x-ray specialist, recognized in Byers' condition symptoms of radium poisoning. Young in years and mentally alert, he could hardly speak. His head was swathed in bandages. He had undergone two successive operations in which his whole upper jaw, excepting two front teeth, and most of his lower jaw had been removed. All the remaining bone tissue of his body was slowly disintegrating, and holes were actually forming in his skull.9
Risks and safety
The early days of radium treatment carried dangers for those handling the radium, many of whom later suffered from radiation-induced cancers; and for patients, who received imprecise doses, often either insufficient to achieve cure; or excessive, causing severe radiation burns; or not sufficiently specifically placed to maximize benefit.

The development of the Geiger counter in 1928, which allowed accurate measurements of radioactivity, and publication of the first international treatment standards by the Christie Radium Institute, Manchester in 1932 were major advances in the early years.
The long half-life of radium, of 1602 years, meant that once purchased, it was endlessly reusable - provided it was not lost, when it would represent a significant very long-term hazard. This was partially addressed by the introduction of radon in the 1940s, sealed into gold tubes. Its half-life of only 3.8 days meant these seeds could be left in place after insertion. However, the challenges of handling a gas that could leak, causing contamination and risks to staff in particular, saw it replaced by short-lived isotopes of gold and iridium (as flexible wires, seeds, needles or removable tapes) from the 1970s.
The use of radium itself died out by the 1980s, being replaced by synthetic radionuclides of shorter half-life, produced in nuclear reactors.
Blue skies success
Before radium, the only treatment for cancer was surgical - a limited option when cancers were often detected late. Chemotherapy was not to develop until the 1940s.9 Today, radiotherapy, chemotherapy and surgery form the three pillars of cancer treatment.
If it was not for Marie Curie's scientific curiosity and the support for her 'blue skies' research, and the recognition of its therapeutic potential, radiotherapy might have taken decades longer to emerge as a key cancer treatment.
Her 1921 speech at Vassar College, reviewing her discovery of radium, makes the same plea for continued funding for basic science that her successors echo today, a century later. One hopes that those funding pure research will pay heed:
We must not forget that when radium was discovered no one knew that it would prove useful in hospitals. The work was one of pure science. And this is a proof that scientific work must not be considered from the point of view of the direct usefulness of it. It must be done for itself, for the beauty of science, and then there is always the chance that a scientific discovery may become like the radium a benefit for humanity.10
Pete Ellis is professor of psychological medicine at the School of Medicine and Health Sciences, University of Otago, Wellington, Wellington, New Zealand.
Alan Dronsfield is emeritus professor of the history of science at the University of Derby.
Acknowledgements
We are particularly grateful to Dr Alan J Gray, retired radiation oncologist, Wellington, New Zealand, for his helpful advice on this paper.
References
- E W Dahlgren, Nobel prize presentation speech, 1911. See http://nobelprize.org/nobel_prizes/chemistry/laureates/1911/press.html (This link will appear in a new window)
- H Becquerel, Comptes Rendus, 1896, 122, 420. See http://web.lemoyne.edu/~giunta/ea/becquerelann.html (This link will appear in a new window)
- Quoted in R F Mould, Current Oncology, 2007, 14, 74. Available (free) at http://www.ncbi.nlm.nih.gov/pmc/articles/PMC1891197/ (This link will appear in a new window)
- J H Sequeira, Brit. Med. Journal, Feb 27, 1915, 365. DOI: 10.1136/bmj.1.2826.365 (This link will appear in a new window)
- J M H MacLeod, Brit. Med. Journal, June 11, 1904, 1366. DOI:10.1136/bmj.1.2267.1366 (This link will appear in a new window)
- R H Herbst, J. Amer. Med. Assoc., 1919, 72, 1610. DOI:10.1001/jama.1919.02610220028010 (This link will appear in a new window)
- Radium-223 is being used to relieve pain in bone cancer. See http://clinicaltrials.gov/ct2/show/NCT00667199 (This link will appear in a new window)
- 'Medicine - radium drinks', Time Magazine, April 11, 1932. See http://www.time.com/time/magazine/article/0,9171,743525,00.html (This link will appear in a new window)
- A T Dronsfield and P M Ellis, Educ. Chem., 2008, 45, 80.
- M Curie, in a lecture given at Vassar College, May 14, 1921.
- R F Mould, A Century of X-rays and Radioactivity in Medicine. London: Taylor and Francis, 1993.
- See http://www.fogonazos.es/2008/06/radio-activity-makes-you-feel-so.html (This link will appear in a new window)









No comments yet