Four unnamed bottles are supplied, each containing a solution of a single compound
Devise experiments to label some bottles correctly, using two different approaches.
Introduction
Teachers who have not used the problems before should read the section Using the problems before starting.
Prior knowledge
Solutions containing aqueous copper(II) ions are blue or blue/green and the use of solubility tables – in particular the insolubility of silver chloride as the basis for the test for chloride ions. A detailed knowledge is unnecessary as students are encouraged to consult textbooks and data books during the exercise.
Resources
Data books and inorganic textbooks should be available for reference. Unnamed, but numbered bottles containing solutions of copper(II) chloride, hydrochloric acid, silver nitrate and sodium nitrate should be provided at the start.
Students in group (i) are not allowed any additional chemicals or flame test equipment, pH paper etc.
Students in group (ii) can request apparatus and chemicals during the practical session, and these should be issued if they are safe to use. In particular, litmus or pH paper, flame test equipment and electrolysis apparatus may be required but they should not be on view.
Special safety precautions
The staining effect of silver nitrate should be noted.
Possible methods
Approach (i)
The blue/green solution is copper(II) chloride. Mix this coloured solution with the other three solutions – the one that gives a precipitate is the silver nitrate. Mix the remaining two with the silver nitrate solution – the one that gives a precipitate is the hydrochloric acid. The remaining solution is the sodium nitrate.
Approach (ii)
- Electrolysis (identifies copper(II) chloride, hydrochloric acid and silver nitrate positively and sodium nitrate by elimination).
- Evaporate each solution to dryness and heat any solid produced (no solid left from HCl, brown gas from silver nitrate, oxygen from sodium nitrate). This method is not recommended in practice because of fumes from the HCl, and because laboratory silver nitrate solution is usually so dilute that there is little solid produced to decompose.
- Other methods include:
- use of pH paper;
- flame tests;
- test for chloride ions with silver nitrate;
- brown ring test for nitrate, but note that the brown ring test may not work with dilute laboratory silver nitrate solution; and
- test for silver ions by adding chloride ions.
A useful strategy
The following strategy could be of help to students using method (i). When handling reactions between a number of substances as in this case, students can find it helpful to write the names of the chemicals in a circle:
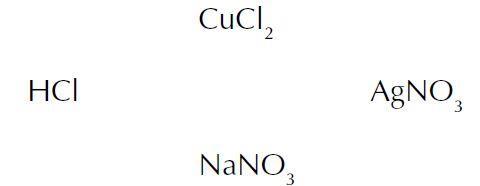
The students choose one of them and draw a line from it to each of the other chemicals in the circle, making notes of the chemical reactions that can occur along each line. This procedure is repeated for the other three compounds.
Suggested approach
During trialling the following instructions were given to students and proved to be extremely effective:
- Divide your group into two subgroups; one of these should use approach (i) and the other approach (ii). The subgroups should move to different parts of the laboratory so that they can work independently.
- Working in your subgroup of two or three, plan how to label the bottles correctly. Write this up in note form. Discussion plays an important part in working out suitable methods. Several minds focusing on a problem together can achieve much more than the same minds working independently.
- Get your subgroup’s method checked for safety and then carry out the practical to find out how well it works. If it doesn’t work properly, try changing it, get it checked and try it out again.
- Write a brief account of what your subgroup did. You should record any changes to your initial scheme and describe any problems you encountered.
- The two subgroups should come together to discuss the two approaches and the advantages and disadvantages of each approach.
- Working as a group, prepare a short (ca 5-minute maximum) presentation to give to the rest of the class. If possible all group members should take part: any method of presentation (such as a blackboard, overhead projector, etc) can be used.
Outline the problem, describe what you did and explain which of the approaches worked best and which you found more interesting. After the presentation, be prepared to accept and answer questions and to discuss what you did with the rest of the class.
Downloads
Four solutions - creative problem solving
PDF, Size 0.15 mb
Additional information
This resource is part of our Creative problem-solving in chemistry collection.


















No comments yet