- Home
- I am a …
- Resources
- Collections
- Remote teaching support
- Starters for ten
- Screen experiments
- Assessment for learning
- Microscale chemistry
- Faces of chemistry
- Classic chemistry experiments
- Nuffield practical collection
- Anecdotes for chemistry teachers
- Literacy in science teaching
- More …
- Climate change and sustainability
- Alchemy
- On this day in chemistry
- Global experiments
- PhET interactive simulations
- Chemistry vignettes
- Context and problem based learning
- Journal of the month
- Chemistry and art
- Classic chemistry demonstrations
- In search of solutions
- In search of more solutions
- Creative problem-solving in chemistry
- Solar spark
- Chemistry for non-specialists
- Health and safety in higher education
- Analytical chemistry introductions
- Exhibition chemistry
- Introductory maths for higher education
- Commercial skills for chemists
- Kitchen chemistry
- Journals how to guides
- Chemistry in health
- Chemistry in sport
- Chemistry in your cupboard
- Chocolate chemistry
- Adnoddau addysgu cemeg Cymraeg
- The chemistry of fireworks
- Festive chemistry
- Collections
- Education in Chemistry
- Teach Chemistry
- Events
- Teacher PD
- Enrichment
- Our work
- More navigation items
In search of more solutions
Try these ideas for chemical "egg race" competitions that engage students and motivate them to follow a scientific or technical career
These resources have been taken from the book In search of more solutions. Aimed at those following post-16 chemistry courses, the book encourages students to design their own experiments in relation to the tasks set, and as they overcome the obstacles within the tasks, their inventiveness and enthusiasm flourishes.
To buy a copy of the book visit our online shop. If you're a member of the Royal Society of Chemistry, you're entitled to a 35% discount.
A chemical stop-clock – iodine clock reaction
Get ready to be exact in your timings, and support learners to create an exact timed reaction. Includes kit list and safety instructions.
A chemically powered boat - a bubble race
An activity worthy of classroom competition, create junk boats, powered by chemistry and sail into learning. Includes kit list and safety instructions.
A hot dinner from a can
Thermochemistry and enthalpy are the topics on the menu for this class experiment. Includes kit list and safety instructions.
A transient red colour: the aqueous chemistry between iron (III) ions and sulfur oxoanions
An exploration into bonding in sulphur oxoanions, and redox reactions. Contains kit list and safety instructions.
After meal puzzle
Use ingenuity and science to solve the riddle of lifting ice with string. Includes kit list and safety instructions.
An elementary problem
Pick the correct chemical process, for the correct day, and figure out when your chemical companies deliver.
As sweet as? Detecting aspartame in a table-top sweetener
Use hydrolysis and chromatography to find aspartame in your table-top sweetener. Includes kit list and safety instructions.
Blanching - what is the most effective method?
Explore enzymes and catalysts in this experiment into fresh food preservation. Includes kit list and safety instructions.
Cool it | In search of more solutions | 14–16 years
Devise a method of reducing the temperature of water to 6.5°C using citric acid and bicarbonate of soda.
Design a pocket handwarmer
Create your own rechargeable exothermic reaction, and keep your fingers toasty with this hand warmer experiment. Includes kit list and safety instructions.
Finding the ‘rate expression’ for the reaction between iodine and tin
Explore rate reaction between two substances, and design an experiment to measure it. Includes kit list and safety instructions.
Fizzy drinks’ froth
Explore hypothesis testing in this experiment using fizzy drinks. Includes kit list and safety instructions
Franklin’s teaspoon of oil
Interpret a historical experiment based on a full and interesting account of Franklin’s contribution to surface chemistry, includes kit list and safety instructions.
From milk to curds and whey - which enzyme?
An experiment, and extension, into enzymes and how they work in dairy products. Includes kit list and safety instructions.
Gas volume
Calculate the gas volume created through the reaction of metals with acids. Includes kit list and safety instructions.
Lifting an egg by a thread
Create a strong cellulose thread and lift and egg. Includes kit list and safety instructions.
Liquid and solid water; the growth of ice crystals
On a cold clear night, the Earth loses heat by radiation. How does a natural pond form ice through the night? Includes kit list and safety instructions.
Making ice | In Search of More Solutions | 11–14 years
Explore the properties of water and ice in this planning activity
Move a heavy object
Use a simple reaction to create CO2, and see how far chemistry can push an object. Contains kit list and safety instructions.
Move an oxo cube at great speed
Make a reaction that creates CO2, and watch as it propels a stock cube faster than any Oxo has travelled before. Includes kit list and safety instructions.
Number of reactions
Explore the chemistry of copper, and weak bases, in this explorative experiment in the reactions of copper(II) sulfate. Includes kit list and safety instructions.
Only dust - is there a sign of life?
Discover the link between organic life, and dust, designing an experiment of your own from junk materials. Includes kit list and safety instructions.
Test the gas
Test or carbon dioxide in this tricky experiment that will make learners consider their scientific skills. Includes kit list and safety instructions.
The candle in the bell jar
How does oxygen, in a sealed container, react when exposed to fire? Find out with this experiment. Includes kit list and safety instructions.
The effect of cooking on vitamin C in cabbage | 16–18 years
Plan a titration experiment to discover more about the changes in vitamins and enzymes during cooking. Includes kit list and safety instructions
The Flatlandian periodic table
Explore a new perspective on chemistry with Flatlandian principals, to create Flatlandia’s periodic table.
Theory v practice - do they compare?
Investigate the reactions of calcium metal, in theory and in practice. Does it act as you’d expect? Includes kit list and safety instructions.
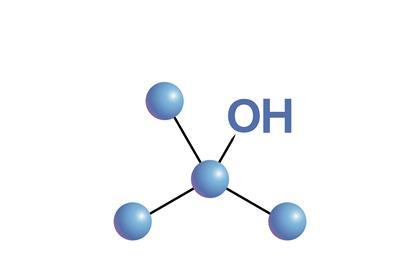
Three isomeric alcohols
Learn about the reactions of primary, secondary and tertiary alcohols, using laboratory tests. Includes kit list and safety instructions.
Vintage titrations: sulfur dioxide concentrations in wine
Explore redox titrations using iodine, and discover the sulfur dioxide concentrations in wine. Includes kit list and safety instructions.
Vintage titrations: tannin in wine
Explore Redox titrations using potassium manganate(VII), with this tannin experiment. Includes kit list and safety instructions.
Warning device
Create a warning system from junk items, and the reaction between citric acid and bicarbonate of soda. Includes kit list and safety instructions.
What is honey made of? The optical rotation of sugars
Discover more about carbohydrates, chirality, optical rotation, in honey. Includes kit list and safety instructions.
What is the smallest amount that you can smell?
Science never smelled better, explore the inner working of smell with this experiment. Includes kit list and safety instructions.
What makes the candle go out?
Why do candles go out, how do different candles react in the same environment, and can we figure out why they’re different? Includes kit list and safety instructions.
Which gas is which?
Five mystery gases, and a problem to solve. Students must identify each gas, and do so quickly. Includes kit list and safety instructions.
Which sodium salt is which?
How does sodium interact with other chemicals, and how can you identify them? Includes kit list and safety instructions.