Explore condensation polymerisation by creating and investigating the properties of a thermosetting polymer in this demonstration
In this experiment, students observe as urea is dissolved in aqueous methanal. By acidifying the solution, the demonstrator can illustrate an example of condensation polymerisation, producing a hard, white, thermosetting polymer. After washing, the properties of this substance can be investigated.
If a mould is used, the experiment can be extended to show the formation of plastic articles made in a mould by condensation polymerisation.
This is a teacher demonstration, taking about five minutes for one polymerisation experiment. Extensions to show production of moulded articles will take longer. It should be performed in a fume cupboard to avoid exposure to methanal vapour.
Equipment
Apparatus
- Eye protection (goggles)
- Disposable nitrile gloves (for handling the polymer)
- Access to a fume cupboard visible to the class
- Measuring cylinder, 100 cm3
- Throwaway containers with secure lids, at least 2 (see note 6 below)
- Dropping pipette
- Glass stirring rod
- Object of simple shape for making suitable mould (see note 7)
- Bunsen burner (optional)
- Tongs (optional)
Chemicals
- Methanal solution (formalin), 37 - 40%, (TOXIC), 20 cm3 (see note 3)
- Urea (carbamide), 10 g
- Sulfuric acid, concentrated (CORROSIVE), 1 cm3 (see note 4)
- Plasticene
- Aluminium foil
Health, safety and technical notes
- Read our standard health and safety guidance.
- Work in a fume cupboard and wear eye protection (goggles) throughout. Wear gloves when handling the polymer.
- Methanal solution (TOXIC) – see CLEAPSS Hazcard HC063. Methanal solution (also known as formaldehyde solution, or formalin) should be in good condition. Because the vapour is unpleasant as well as toxic, the supply of this solution for this demonstration should be kept in the fume cupboard, in a stoppered bottle.
- Concentrated sulfuric acid (CORROSIVE) – see CLEAPSS Hazcard HC098a. The acid used should be sulfuric acid, and NOT hydrochloric acid, because of the possibility of forming the carcinogen, bis-chloromethylether.
- Urea – see CLEAPSS Hazcard HC035A.
- The container used should be transparent (so the demonstration can be seen), disposable, and preferably should have a secure lid. Used 100 g coffee jars with screw tops are ideal, but any similar size glass or plastic container will do.
- A mould for casting copies of an object can be made by pressing the object into Plasticine. The object selected should have a simple external shape. Enough Plasticine should be provided to press the shape into, forming a sufficiently substantial mould to retain its shape when handled.
- After the demonstration, dispose of the sealed container with the polymer inside as solid waste.
Procedure
- Dissolve 10 g of urea in 20 cm3 of methanal solution in the disposable container.
- Add about 1 cm3 of concentrated sulfuric acid a drop at a time, using a dropping pipette, and stir steadily. Within a minute the solution begins to go milky and eventually a hard, white solid is formed which is difficult to remove from the container. A lot of heat is evolved.
- Show that the polymer is hard by poking the material with a spatula.
- Wash the polymer thoroughly before passing around the class, as it is likely to be contaminated with unreacted starting materials. Alternatively, pass it round in the container with the lid screwed on.
- Make a plasticene mould from the simple shape and line it with aluminium foil.
- Make another urea-methanal solution as above, but, immediately after adding the acid, pour some of the solution into the mould and allow it to polymerise. Remove from the mould when solid.
- Hold a sample of the polymer (from the mould) with tongs and heat in a Bunsen flame. It will char but not melt, showing that it is a thermosetting polymer.
Teaching notes
The reaction is a condensation polymerisation in which water is eliminated as the hydrogen atoms from the ends of one amino-group from each of two urea molecules combine with the oxygen atom from a methanal molecule. The remaining –CH2– group from the methanal molecule then forms a bridge between two neighbouring urea molecules, as shown below. This process, repeated many thousands of times, forms long chains of urea and methanal molecules linked in this way.
Sometimes the second hydrogen atom on an amino-group will also react with a methanal molecule, producing a branch in the chain, and chains may even become cross-linked to each other. Eventually a random three-dimensional network of cross-linked chains is formed, giving a rigid structure and thus a hard, inflexible material.
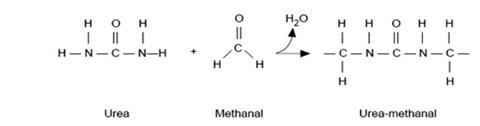
The product has many cross links as shown in the diagram below.
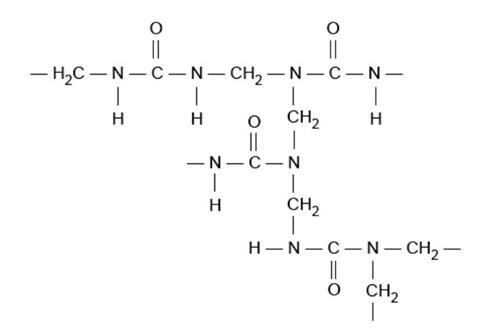
Because the tangle of cross-linked chains is almost impossible to separate, the material does not melt on heating, although it will eventually break down at high temperature, decomposing and giving off small molecules such as steam and nitrogen, leaving a charred mass which is largely carbon.
Additional information
This is a resource from the Practical Chemistry project, developed by the Nuffield Foundation and the Royal Society of Chemistry.
Practical Chemistry activities accompany Practical Physics and Practical Biology.
© Nuffield Foundation and the Royal Society of Chemistry


















No comments yet