Strategies to help your students master reactant and product ratios and calculate reacting and product masses
Within chemical industries, understanding reacting masses is fundamental to maximising product output while minimising waste. From making industrial-scale fertilisers, such as ammonium nitrate, to preparing everyday medicines like aspirin or paracetamol in pharmaceutical labs, chemists use reacting mass calculations to work out how much of each substance they need. These industrial processes demonstrate how balanced equations become practical tools that help predict and control the amounts of reactants and products.
Introducing reacting masses to students helps them see how the quantities in a balanced equation relate directly to the amounts of substances used and made in a reaction. This is the point when learners begin to connect the symbolic equations to real amounts, and this allows them to make straightforward, quantitative predictions about chemical change.
What students need to know
At 11–14, students should know that a chemical reaction involves a rearrangement of atoms in which the reactants form products and be able to represent reactions using word equations. Students may even have been introduced to simple balanced symbol equations and have begun to understand that substances combine in fixed ratios. Ensure your 14–16 learners know that:
- Mass is conserved in a chemical reaction.
- A balanced symbol equation gives the ratio in which substances react or are produced.
- The relative formula mass of a substance in grams contains one mole or 6.02 x 1023 particles.
- Reacting masses can be calculated by using either the mass-ratio method or the mole method.
- The maximum amount of product that can be formed is determined by the limiting reactant.
Misconceptions
Misunderstandings in this topic frequently stem from deeper difficulties students experience with stoichiometry. Learners often struggle with the concept of the mole, by viewing it as an abstract or arbitrary number rather than a precise way of counting particles. This can prevent students from understanding why balanced equations provide the ratios needed for quantitative work.

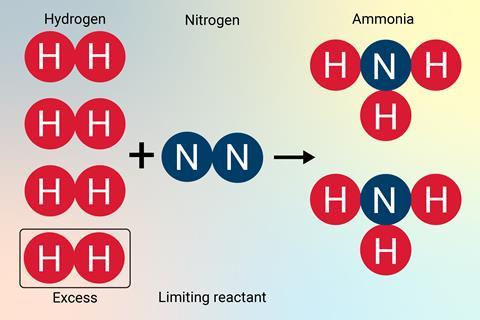
Another misconception is that the number of moles is conserved during a reaction; for example, if 0.5 mol of nitrogen reacts with 1.5 mol of hydrogen to form ammonia, they expect a total of 2.0 mol of product rather than 1.0 mol. Students need a secure understanding of moles as a way of counting particles to overcome this.
Identifying the limiting reactant can also be challenging. Students may assume that the reactant with the smaller amount in moles must be limiting, rather than comparing the amounts relative to the stoichiometric numbers.
A useful model is to pretend to make a sandwich with representative pieces of coloured card (bread = white card, tomato = red card, cheese = yellow card). Every sandwich has two slices of bread, three slices of tomato and one slice of cheese: (2B + 3T + C → B2T3C). As students make the sandwiches, they see the ratio of 2:3:1, modelling ratios within balanced symbol equations. Now give 10 slices of bread, 5 slices of cheese, but only 12 slices of tomato. Students quickly see that tomatoes, not the fewest items, limit the number of sandwiches that can be made.
What you need to know
Quantitative chemistry is an area in which teachers can unintentionally experience a degree of expert blindness because the steps feel logical and intuitive. Be aware that each additional step competes for space in students’ working memory. Teacher modelling is a common and effective way to teach quantitative chemistry, but to scaffold the process effectively, we must delay the handover to help break the process into manageable chunks.
- Break calculations into small, incremental steps. Build on prior knowledge and check understanding at each stage before moving on.
- Use tables to help structure multi‑step calculations. A consistent layout reduces the load on working memory for students and helps them see the link between each stage of the process.
- Take time to introduce the mole. Emphasise that it represents an amount of substance rather than a formula to memorise to prevent misconceptions.
- Help students understand limiting reactants through a simple model, such as making a sandwich. Then give them a clear method to identify the limiting reactant during reacting‑mass calculations.
Suggestions for your teaching
When students first attempt reacting mass questions, start with the mass‑ratio method to reduce cognitive load because it builds on ratio and proportion skills that should be familiar from maths. The balanced symbol equation gives the particle ratio, but it should be made explicit that this alone does not tell us the masses of the substances involved because different particles have different masses.
To calculate reacting masses, convert the particle ratio into a mass ratio. Provide students with a table to break the calculation down into manageable steps and scaffold the learning. This reduces the demand on working memory and allows students to focus on the underlying chemical reasoning.
Calculate the mass of magnesium oxide produced when 12g of magnesium is burned in excess oxygen.
| 2Mg + O2 → 2MgO | ||||
|---|---|---|---|---|
|
Row 1: Enter the information from the question. |
Mass given in question |
12g |
||
|
Row 2: Calculate the relative atomic/formula mass for each substance. (Remember: the stoichiometric numbers do not affect this.) |
Relative atomic/formula mass |
24 |
32 |
40 |
|
Row 3 : Extract the stoichiometric ratio from the equation. |
Stoichiometric ratio |
2 |
1 |
2 |
|
Row 4: Multiply the stoichiometric ratio by the relative atomic/formula mass to give the mass ratio and check that the total mass of the reactants equals the total mass of the products. |
Mass ratio |
48g |
32g |
80g |
|
Row 5: Use the mass ratio to answer the question. (If students struggle with this conversion, you can add in an extra row that scales down to 1g first, before scaling up to the answer.) |
Use this ratio |
12g |
8g |
20g |
Although the mass-ratio method is a logical progression and sufficient for many reacting mass questions at this level, the introduction of the mole as an amount of substance allows learners to answer questions that involve limiting reactants. It also prepares them for post‑16 work involving concentration and gas volumes.
The mole can be a concept that learners struggle to comprehend, so it is important that they are clear that a mole of a substance signifies a specific number of particles. Much like a dozen eggs or red roses signifies 12, a mole signifies 6.02 x 1023 particles.
Once students are confident with converting between a mass and an amount in moles, they can incorporate this into calculating reacting masses. Questions that involve only one given mass are often straightforward, but identifying the limiting reactant can add challenge.
Once students have calculated the amount in moles of each reactant, direct them to divide each value by its stoichiometric number from the balanced equation. The smaller number identifies the limiting reactant. This helps avoid the misconception that the smallest mass or smallest amount in moles must be limiting and instead focuses attention on comparing reactants in the correct ratio.
If 22.4 g of nitrogen reacts with 4.50g of hydrogen, what mass of ammonia is produced?
| N2 + 3H2 → 2NH3 | ||||
|---|---|---|---|---|
|
Row 1: Enter the information from the question. |
Mass given in question |
22.4g |
4.50g |
|
|
Row 2: Calculate the relative atomic/formula mass for each substance. (Remember: the stoichiometric numbers do not affect this.) |
Relative atomic/formula mass |
28 |
2 |
17 |
|
Row 3: Calculate the amount in moles. |
Moles |
0.80 |
2.25 |
|
|
Row 4 : Extract the stoichiometric ratio from the equation. |
Stoichiometric ratio |
1 |
3 |
2 |
|
Row 5: Check which reactant is limiting by dividing the amount in moles by the stoichiometric number. |
Limiting reactant |
0.80 |
0.75 |
|
|
Row 6: Use the stoichiometric ratio to determine the amount in moles of desired reactant/product based on limiting reactant. |
Use the stoichiometric ratio to determine moles |
2.25 |
1.5 |
|
|
Row 7: Convert the amount in moles to a mass. |
Calculate final answer |
25.5g |
||
Encourage students to construct their own tables by providing them with strips with the first column that they can place beside each question to support as needed.
Reacting masses offer students a way to connect the symbolic equations they learn with the measurable quantities they encounter in the laboratory. By breaking the topic into small steps and making our thinking explicit, we can support learners to navigate the challenges of quantitative chemistry. This careful approach helps them develop confidence, avoid common misconceptions and build the foundations they will need for further study.
Resources for your classroom
- Deepen your learners understanding of ratios in balanced chemical equations with developing understanding worksheets.
- Cater to all learners with these differentiated worksheets designed for learners to review their learning.
- Help students feel more comfortable with concepts, such as moles, ratios and reagents, with this cooking equations worksheet.
- Discover everything you need to teach quantitative chemistry at 14–16, including classroom activities, experiments and top teaching tips, in our topic package.
Resources for your classroom
- Deepen your learners understanding of ratios in balanced chemical equations with developing understanding worksheets: rsc.l/wheelbarrow
- Cater to all learners with these differentiated worksheets designed for learners to review their learning: rsc.l/wheelbarrow
- Help students feel more comfortable with concepts, such as moles, ratios and reagents, with this cooking equations worksheet: rsc.l/wheelbarrow
- Discover everything you need to teach quantitative chemistry at 14–16, including classroom activities, experiments and top teaching tips, in our topic package: rsc.li/wheelbarrow














No comments yet