The Thermite in a flowerpot demonstration generates a large quantity of smoke, which means it isn’t suitable for all laboratories. This microscale version can be used when no suitable outdoor space or laboratory is available for the larger scale version. The reaction is on a much smaller scale and takes place under a beaker – perfect for containing the mess but keeping the magic.
Get started
Watch a demonstration of this experiment and download the technician notes from the Education in Chemistry website: rsc.li/4w44o4u
If your lab has smoke detectors or less than ideal ventilation, Thermite in a flowerpot might produce too much smoke for you. Use this microscale thermite demonstration instead. The reaction is on a much smaller scale and is contained under a beaker – perfect for containing the mess but keeping the magic.
Kit
- Heat resistant mats (or a piece of hardboard)
- Sand
- Small tin with wide base (e.g. a salmon or sardine tin)
- Small filter paper (e.g. 4.5 cm diameter)
- 0.25 g aluminium powder (fine or medium)
- 0.75 g iron(III) oxide powder
- 0.05 g magnesium powder
- 0.45 g barium nitrate(V)
- 6 cm magnesium ribbon
- 1 L borosilicate beaker
- Bunsen burner
Preparation
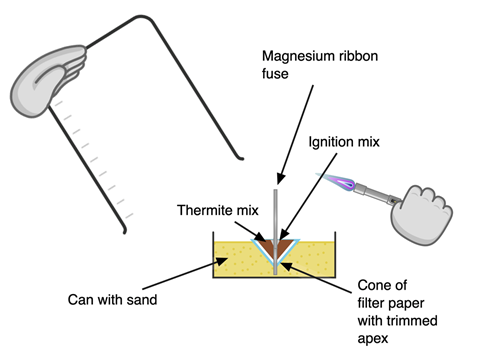
Clear the desk of combustibles and use heatproof mats (or a piece of hardboard) to cover the bench. Fill the tin with sand to about 3 cm depth. Place it on a mat. Fold the filter paper to make a cone and trim the end to make a hole just wide enough for a piece of magnesium ribbon to fit through. Stick the cone into the sand.
Make 1 g of thermite powder by combining 0.25 g of aluminium powder with 0.75 g of iron(III) oxide. Make no more than 0.5 g of ignition powder by mixing 0.45 g of barium nitrate(V) with 0.05 g magnesium powder. When weighing the powders, avoid flames, raising dust and skin contact. Never stir the mixtures. Create mixtures by carefully transferring the powders from one filter paper to another and back again. The ignition mixture is particularly sensitive to friction and impact. Make fresh powder mixtures for each demonstration.
Pour the thermite mixture into the filter paper cone. Make a small depression in the top of the thermite mixture for the ignition mixture and pour this on. Cut 6 cm of magnesium ribbon and feather one end by cutting slits into it.
In front of the class
Position the learners 4 m away and ensure they’re wearing eye protection and protected by a safety screen. Instruct students not to look directly at the reaction, except through a passive welding filter, shade 9.
Push the unfeathered end of the magnesium ribbon fuse through the ignition mixture, thermite mixture, filter paper and sand to touch the base of the can. Ensure there is good contact between the fuse and the powders.
Hold the 1 L beaker in one hand and a lit Bunsen in the other. Light the magnesium ribbon, cover the equipment with the beaker and stand back.
When the fuse hits the ignition mix, a bright flash follows and the reaction glows white hot. If the mixture does not light first time, wait a few minutes in case of a delayed reaction before disposing of it. CLEAPSS members can consult L195 which outlines potential fixes for reactions that do not work (bit.ly/4dbHgt3). Once the reaction has cooled, remove the beaker and collect the iron with a magnet.
When the fuse hits the ignition mix, a bright flash follows and the reaction glows white hot. If the mixture does not light first time, wait a few minutes in case of a delayed reaction before disposing of it. CLEAPSS members can consult L195 which outlines potential fixes for reactions that do not work. Once the reaction has cooled, remove the beaker and collect the iron with a magnet.
Teaching goal
Although the thermite reaction is commonly demonstrated to learners aged 14–16, it’s also worth returning to it at post-16 level because it has a very accessible enthalpy of reaction calculation.
| Compound | ∆Hf (kJ mol-1) |
|---|---|
| Fe2O3 | –825.50 |
| Al2O3 | –675.7 |
The thermite reaction has an enthalpy of reaction of about –850 kJ mol-1. How does this compare with other reactions? Does the drama of the reaction reflect a particularly large change in enthalpy?
| Reaction | ∆Hr (kJ mol-1) | Balanced equation | Molar mass of reactants | Energy density (kJ g-1) |
|---|---|---|---|---|
| Combustion of carbon | –394 | C + O2 → CO2 | 12 + 32 = 44 g mol-1 |
394 kJ mol-1 / 44 g mol-1 = 9.0 kJ g-1 |
| Thermite | –850 | Fe2O3 + 2 Al → Al2O3 + 2 Fe |
(2×27.0) + 159.7 = 214 g mol-1 | 850 kJ mol-1 / 214 g mol-1 = 4.0 kJ g-1 |
If you compare this value with the enthalpy of combustion of carbon (graphite), which is 394 kJ mol-1, you might be tempted to say ‘yes’. But when you consider the molar masses involved, for all its apparent ferocity, the thermite reaction actually releases around half the energy per gram of reactant than a barbeque does.
This suggests a kinetic effect. This same phenomenon explains the dramatic sodium and potassium with water reactions (despite them releasing less energy than lithium), and the screaming jelly baby demonstration (compared to a burning marshmallow) (rsc.li/3OKmckf).
This suggests a kinetic effect. This same phenomenon explains the dramatic sodium and potassium with water reactions (despite them releasing less energy than lithium), and the screaming jelly baby demonstration (compared to a burning marshmallow).
The low density of the mixture (typically around 0.7 g cm-3 – although this can be higher with compressed formulations) means a little input of heat can greatly increase its temperature.
The thermite reaction rapidly reaches aluminium’s melting point. While its boiling point (2519°C) is comparable to first-row d-block elements such as iron or copper, its melting point (660°C) is significantly lower. This allows what would otherwise be a solid–solid reaction to get a kinetic boost as the aluminium melts.
While a barbeque needs oxygen from the atmosphere, the iron(III) oxide mixed through the melting aluminium acts as an oxidant. This means the reaction can continue to heat up and accelerate beyond the melting point of iron (1538°C) all the way up to iron’s boiling point (2861°C), provided the reaction scale is large enough.
Health, safety and disposal
- Read our standard health and safety guidance and carry out a risk assessment before running a live demonstration (rsc.li/4mO9EVF). Consult your local safety advisory body for specific guidance. CLEAPSS members should consult L195 which this method follows. Do not deviate from the instructions or attempt to scale up the reaction.
- Always wear eye protection and a lab coat.
- The aluminium powder, magnesium powder and magnesium ribbon are all flammable. The barium nitrate (V) is an oxidiser that is harmful if inhaled or ingested.
- Dispose of the reaction remnants in the normal bin. Unreacted powder requires treating first. Pour it into a beaker of dilute 1M or 2M hydrochloric acid or sulfuric acid and leave overnight in a fume cupboard. Check the next day that the remaining solution is still acidic. Collect any remaining solid by filtration, double bag it and put it in the bin. Pour the solution down a foul water drain with further dilution.
Health, safety and disposal
- Read our standard health and safety guidance and carry out a risk assessment before running a live demonstration. Consult your local safety advisory body for specific guidance. CLEAPSS members should consult L195 which this method follows. Do not deviate from the instructions or attempt to scale up the reaction.
- Always wear eye protection and a lab coat.
- The aluminium powder, magnesium powder and magnesium ribbon are all flammable. The barium nitrate (V) is an oxidiser that is harmful if inhaled or ingested.
- Dispose of the reaction remnants in the normal bin. Unreacted powder requires treating first. Pour it into a beaker of dilute 1M or 2M hydrochloric acid or sulfuric acid and leave overnight in a fume cupboard. Check the next day that the remaining solution is still acidic. Collect any remaining solid by filtration, double bag it and put it in the bin. Pour the solution down a foul water drain with further dilution.
Downloads
Microscale thermite technician notes
Handout | PDF, Size 0.29 mbMicroscale thermite technician notes
Editable handout | Word, Size 0.58 mb





















No comments yet