A Professor has invented a ‘portable chemical circuit breaker’ which when activated breaks an electrical circuit after a time delay of two minutes
However, she can’t remember how she made it, what it could be used for, or even where her notebook is. Can your learners help?
This experiment should take 170 minutes (this time can be split into a 30-minute planning lesson where students have all the apparatus available to look at, and two 70-minute practical sessions).
Task
Help the Professor reconstruct her device (remember it must break the circuit two minutes after being activated) and suggest what it might be used for?
Your device must be ready for operation at the end of the practical and will be activated when the Professor says so. Once activated, the circuit should then be broken without you having to touch your device any more.
Evidence of circuit break is to be clearly visible.
NB A chemical reaction must be involved - A diagram of your invention, with some explanation, will be needed.
Equipment
- Eye protection
- Junk items
- Filter paper
- Rubber and glass tubing of various sizes
- Small pieces of polythene sheeting
- Plastic syringes
- Stop clocks
- Small electric bulb
- Electric battery (dry cell) suitable for use with light bulb
- Copper wire SWG 28, 35 cm
- Zinc foil, 1.5 g
- Magnesium ribbon, 2 g
- Sulphuric acid, 1 mol dm–3, 300 cm3
- Copper(lI) sulfate as “catalyst” (to enable zinc to be used).
- Depending on the knowledge of students, it might be better to treat the zinc with dilute copper sulfate solution first
Health, safety and technical notes
- Read our standard health and safety guidance here.
- Wear eye protection.
- This is an open-ended problem-solving activity, so the guidance given here is necessarily incomplete.
- Students should be told not to concentrate the acid provided.
- Teachers need to be vigilant that groups do not generate gas in a sealed container (other than a syringe, plastic bag or similar) because of the risk of a pressure build up/spraying acid around.
- Sulfuric acid, 1 mol dm–3 H2SO4(aq), is an irritant, see CLEAPSS Hazcard HC098a
- Zinc powder, Zn (s) is flammable and hazardous to the aquatic environment, see CLEAPSS Hazcard HC107.
- Magnesium ribbon, Mg (s) is flammable, see CLEAPSS Hazcard HC059b.
- Copper(II) sulfate solution, CuSO4 (aq) causes eye damage, is harmful if ingested and is hazardous to the aquatic environment, see CLEAPSS Hazcard HC027c.
- Hydrogen gas is highly flammable, and hydrogen/air mixtures can be explosive. Keep away from sources of ignition.
Possible approaches
There is no obvious best method. Approaches already noted:
- Reservoir of sulphuric acid dripping onto a magnesium fuse, calibrated so that the magnesium dissolves after required time. Varying accuracy depending on construction.
- Magnesium/acid reaction used to generate hydrogen. Hydrogen inflated balloon and movement of balloon breaks circuit contact. Variations on this method were most successful.
- Magnesium/acid reaction used to generate hydrogen. Hydrogen production moves a syringe which operates a circuit breaker. Combination of acid concentration and distance moved by plunger used to vary time. Times variable depending on neatness of construction. All those adopting this method were short of time.
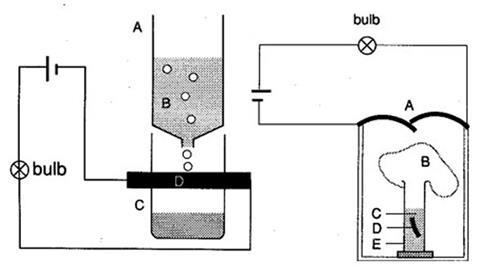
- Left diagram: A = Top of plastic washing up bottle. B = Acid reservoir. C = Bottom of plastic washing up bottle. D =Magnesium ribbon contacts.
- Right diagram: A = Magnesium ribbon contacts. B = Balloon. C = Acid reservoir. D = Magnesium ribbon. E = Glass tube.
Possible extension
Some students may wish to refine their device in their own time.
Notes
Zinc and copper are harmful for the environment. Solutions containing these should be kept for disposal. (It may be easier to precipitate as the carbonate, filter and store the solid). It is the responsibility of the teacher to carry out a suitable risk assessment.
Downloads
Chemical circuit breaker
Experiment | PDF, Size 51.07 kb
Additional information
The resources were originally published in the book In Search of Solution P. Borrows, K. Davies and R. Lewin, Royal Society of Chemistry, 1990.
This experiment was based on an idea contributed by R.F. Kempa.


















No comments yet