This resource from the interactive lab primer highlights the vocabulary and standards most commonly used in university and research chemistry laboratories.
Introduction
This resource highlights the vocabulary and standards most commonly used in university and research chemistry laboratories.
Contents
Abbreviations and physical constants
Common conversions and multiplication factors
Abbreviations and physical constants
Abbreviations
There are many abbreviations you will meet in laboratory situations. Here is a list of the most common and more useful abbreviations.
Abbreviation |
Meaning |
|
Ar b.pt. COSHH DCM EDTA en F FT-IR GC HASAW HPLC IR IUPAC Ka Ks Kw Mr m.pt. NMR PTFE R Rf SI STP TCA TLC TMS UV |
Relative Atomic Mass Boiling Point Control of Substances Hazardous to Health Dichloromethane Ethylenediaminetetraacetic Acid Ethylenediamine Faraday Constant Fourier Transform Infrared (Spectroscopy) Gas Chromatography Health and Safety at Work High Performance Liquid Chromatography Infra Red International Union of Pure and Applied Chemistry Acid Dissociation Constant Solubilty Product Ionic Product of Water Relative Molecular Mass Melting Point Nuclear Magnetic Resonance Polytetrafluoroethylene (e.g Teflon) Universal Gas Constant Retention Factor System Internationale d’Unites Standard Temperature and Pressure Trichloroacetic Acid Thin Layer Chromatography Tetramethylsilane Ultraviolet |
Physical constants
The use of physical constants will be inevitable in practical write-ups where calculations are required. Here are some of the more common ones.
Term |
Symbol |
Value |
|
Avogadro Constant Molar Gas Constant Faraday Constant Molar Volume of an Ideal Gas at 100kPa and 273K Atomic Mass Unit |
L, NA R F VM a.m.u. |
6.022 x 1023 mol-1 8.314 J K-1 mol-1 9.649 x 104 C mol-1 22.7 dm3 1.661 x 10-27 kg |
Common conversions and multiplication factors
Common conversions
Converting from one unit to a different one can cause students problems. Here are some of the most common conversions that are often necessary.
Mass conversions
|
÷ 1000 |
÷ 1000 |
÷ 1000 |
||||
|
microgram(µg) |
|
milligram(mg) |
|
gram (g) |
|
kilogram (kg) |
|
|
|
|
||||
|
x 1000 |
x 1000 |
x 1000 |
Volume conversion
|
÷ 1000 |
÷ 1000 |
|||
|
cm3 |
|
dm3 |
|
m3 |
|
|
|
|||
|
x 1000 |
x 1000 |
Temperature conversion
|
+273 |
||
|
oC |
|
K |
|
|
||
|
-273 |
Multiplication factors
In chemistry you have to deal with numbers of a wide range of magnitudes and for convenience these can be expressed as multiplication factors such as ‘kilo’ or ‘micro’. A complete range is shown here.
Name |
Multiplication factor |
Symbol |
|
tera giga mega kilo deci centi milli micro nano pico femto |
1012 109 106 103 10-1 10-2 10-3 10-6 10-9 10-12 10-15 |
T G M k d c m µ n p f |
Other sections
Return to the interactive lab primer home

Additional information
The interactive lab primer was developed as part of the ‘Chemistry for our future’ Royal Society of Chemistry Teacher Fellowship Scheme. With contributions from Prof. David Read (the University of Southampton), Mr David Brentnall (The University of Nottingham), Dr Sandy Wilkinson (the University of Birmingham) and Mr William Davey (The University of Sheffield)
© Royal Society of Chemistry












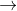






No comments yet