Five teaching tips that ensure your learners have a solid understanding of this simple but fundamental topic

We teach and assess testing for gases in both the 11–14 and 14–16 stages of chemistry education. It’s a relatively simple topic requiring recall of gas test methods and observations for a positive test. For most students, we limit this to hydrogen, oxygen, carbon dioxide and chlorine. However, if students are going to retain their knowledge, we need to help them create strong memory associations.
1. Put safety first
Working with gases – and teenagers – is not a trivial matter, and there are a lot of factors you need to take into account. Hydrogen gas is flammable, oxygen gas is oxidising, chlorine gas is toxic and carbon dioxide is harmful. Check the relevant safety sheets to make sure what you are planning is appropriate for the age and stage of your students, and ensure the lab is well ventilated to reduce the risks that come with the whole class working with gases. It is important to consider where you will place the Bunsen burners as well as how you will manage students walking around with lit splints and flammable chemicals.
1. Safety first
Working with gases – and teenagers – is not a trivial matter, and there are a lot of factors you need to take into account. Hydrogen gas is flammable, oxygen gas is oxidising, chlorine gas is toxic and carbon dioxide is harmful. Check the relevant safety sheets to make sure what you are planning is appropriate for the age and stage of your students, and ensure the lab is well ventilated to reduce the risks that come with the whole class working with gases. It is important to consider where you will place the Bunsen burners as well as how you will manage students walking around with lit splints and flammable chemicals.
2. Avoid assumptions
If learners need to collect gases in test tubes, check they understand what to do to reduce the unnecessary escape of gases. Whether this is simply putting a thumb over the top of a test tube, bubbling the gas directly into limewater or collecting a tube of gas over water, don’t assume they have the necessary skills or confidence. Ensuring they grasp this important information will also help the lessons run more smoothly.
3. Simplify practicals
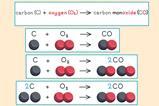
If students need to generate the gases for testing, they will need to do a chemical reaction. This can add to the cognitive load of the lesson, so consider ways you can reduce the number of steps (rsc.li/4sUN9QG). For example, if you’re using zinc or magnesium to generate hydrogen gas from acid, could you measure the metal out and put it in test tubes before the lesson? Technicians can be an excellent source of advice in this area.
3. Simplify practicals
If students need to generate the gases for testing, they will need to do a chemical reaction. This can add to the cognitive load of the lesson, so consider ways you can reduce the number of steps. For example, if you’re using zinc or magnesium to generate hydrogen gas from acid, could you measure the metal out and put it in test tubes before the lesson? Technicians can be an excellent source of advice in this area.
4. Check the language
Most examination specifications are happy with simple language such as ‘lit splint, squeaky pop’ for the test for hydrogen and its result. The results of the test for carbon dioxide can be trickier. Many resources describe the result as limewater turns ‘milky’ or ‘cloudy’, but some exam boards don’t allow this and require ‘white precipitate’. The test for chlorine is another where examiners can be quite strict about the language learners use. Check recent mark schemes for pointers.
5. Focus on knowledge
Teach the gas tests as the sole aim of a lesson – it will place the key knowledge front and centre. Follow up with a quiz in the next lesson to check how well students recall the facts, then look for opportunities to reinforce the knowledge in context. Lots of reactions produce gases. Even if you don’t have time for learners to test them within their practical work, you can always include a question in the follow up on how they could confirm the identity of the gas. Gases are also produced during electrolysis and when studying combustion, such as when learning organic chemistry topics. The more often you aim to prompt recall of this in your lessons, the better.
For older students, drop in some recall questions on gas tests in lessons on gas calculations and easy verbal quizzing whenever they encounter an equation with a gas. By the time you get to the final exams, students should be secure with these key facts.
5. Keep knowledge in focus
Teach the gas tests as the sole aim of a lesson – it will place the key knowledge front and centre. Follow up with a quiz in the next lesson to check how well students recall the facts, then look for opportunities to reinforce the knowledge in context. Lots of reactions produce gases. Even if you don’t have time for learners to test them within their practical work, you can always include a question in the follow up on how they could confirm the identity of the gas. Gases are also produced during electrolysis and when studying combustion, such as when learning organic chemistry topics. The more often you aim to prompt recall of this in your lessons, the better.
For older students, drop in some recall questions on gas tests in lessons on gas calculations and easy verbal quizzing whenever they encounter an equation with a gas. By the time you get to the final exams, students should be secure with these key facts.
Kristy Turner
More resources
- Develop your 14–16 learners’ independent writing skills with this reacting masses and gas volumes structure strip.
- Use these classroom activities and tips when teaching gases at 14–16.
- Once your students are solid in their understanding, put their knowledge to the test with this gas test classroom activity.
- Encourage learners to assess risk effectively and stay safe during practical work with this Teaching practical science article.













No comments yet