Use this demonstration with your 14–16 learners to deduce the formula of copper(II) oxide by reducing it with methane
-

Download this
Download the scaffolded and unscaffolded student worksheets and teacher guidance (including answers) as MS Word and pdf and the presentation slides as MS Powerpoint and pdf.
Discover more resources from the Nuffield practical collection
In this experiment, learners heat copper(II) oxide in a glass tube while passing methane over it, reducing the copper(II) oxide to copper. If they measure the mass of the reactants and products carefully, learners can then deduce the formula of the copper oxide. This can also be used simply as an example of reduction.
Always demonstrate this experiment to learners before they attempt it themselves, so you can highlight all the associated risks and the safety precautions that need to be taken.
The practical will take up to an hour, perhaps more to analyse the results. It is not suitable as a class practical for learners under the age of 14 but is a useful demonstration.
Learning objectives
- Safely heat copper oxide with methane to reduce copper oxide to copper metal.
- Accurately record the change in mass.
- Explain the observed mass changes.
- Calculate the relative formula mass using experimental data.
Success criteria
- Use the mass balance correctly to record masses by using the tare function correctly (LO2).
- Safely heat the glass tube and burn the excess methane (LO1).
- Explain mass change by linking to knowledge of conservation of mass and balanced equations (LO3).
- Link knowledge of conservation of mass with moles to calculate the relative formula mass (LO4).
Scaffolding
As mentioned above, this practical is not easy to do, so consider doing the experiment only as a demo if, having considered the risks, you feel this is not appropriate for your group.
There is a scaffolded and unscaffolded version of the question sheet. If calculating moles is not something you would teach to some classes, the scaffolded student sheet allows the key points to be explored anyway.
Answers to the student worksheet questions are at the end of these teacher notes.
Equipment
Apparatus
- Safety glasses
- Reduction tube (hard glass test tube with small hole near closed end)
- One-hole bung with glass tube to fit the reduction tube
- Rubber tubing
- Clamp stand, boss and clamp
- Bunsen burner
- Heat resistant mat
- Spatula
- Balance (must be accurate to at least 0.01 g)
Chemicals
- Copper(II) oxide, 2 spatulas (HARMFUL, DANGEROUS FOR THE ENVIRONMENT). For best results, use analytical grade copper(II) oxide which has been dried by heating in an open dish at 300–400°C for 10 minutes and then stored in a desiccator.
- Methane (natural gas) (EXTREMELY FLAMMABLE). It is likely that most schools will use a class gas tap for this experiment. This gas forms explosive mixtures in the air.
Health, safety and technical notes
- Read our standard health and safety guidance.
- Wear safety glasses throughout.
- Copper(II) oxide, CuO(s), (HARMFUL, DANGEROUS FOR THE ENVIRONMENT) – see CLEAPSS Hazcard HC026, refer to SSERC or contact your local safety advisory body.
- Methane (natural gas), CH4(g), (EXTREMELY FLAMMABLE) – see CLEAPSS Hazcard HC045a, refer to SSERC or contact your local safety advisory body.
Method
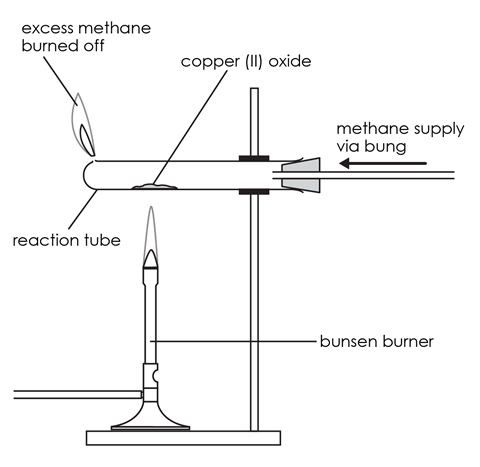
- Measure the mass of the test tube with the bung in (mass 1). Put 2 spatulas of copper(II) oxide into the tube and spread it out as much as possible.
- Measure the mass of the tube again, with the copper oxide in it (mass 2).
- Assemble the apparatus as shown in the diagram, but do not place the Bunsen burner underneath yet. Clamp the test tube as near to the bung as possible.
- Turn on the gas tap attached to the test tube about halfway to get a steady flow of gas. This will pass methane through the apparatus.
- Light the gas coming out of the hole at the end of the tube. Your teacher should supervise this step. Take care not to lean over the tube as you light the gas. Adjust the gas tap so that the flame is about 3 cm high.
- Light the Bunsen burner and begin to heat the copper oxide in the tube. You will need to use a roaring flame (air hole fully open). Pick up the Bunsen burner and move the flame around to heat every bit of the copper oxide. Make sure that you use the hottest part of the Bunsen burner flame (the top of the inner cone) for heating. If there are parts which look unreacted, gently shake the tube – it will be very hot, so do so by gently shaking the clamp stand.
- When all the copper oxide looks like it has reacted (it will look like copper), keep heating for a further minute or two and then turn off the Bunsen burner.
- Keep the methane passing over the product as it cools down to prevent it from reacting with any oxygen present and turning back into copper oxide. When the tube is cool, switch off the gas.
- Measure the mass of the test tube with the bung and the product (mass 3).
Teaching notes
Learners should complete the results table below throughout the practical. While this practical is focused on the mass and how this changes, ask learners to also write observations as the copper oxide (black) changes colour when it is reduced to copper (reddish/orange). Make sure that learners heat the copper oxide until all the copper is this colour. Asking questions about the colour and why this is important will help learners with some of the later questions on their sheet.
Questions linking the practical experiment to quantitative chemistry topics can be found in the student worksheets. There are two versions of the student worksheet: scaffolded and unscaffolded. The scaffolded sheet offers more support to allow learners to access the questions. Hints are provided after some of the questions to support learners and guide their answers.
Answers
Answers to the questions in both levels of student sheets and on the lesson slides can be found in the teacher notes.
Downloads
Find the formula of CuO scaffolded student sheet
Handout | PDF, Size 0.28 mbFind the formula of CuO unscaffolded student sheet
Handout | PDF, Size 0.26 mbFinding the formula of CuO slides
Presentation | PDF, Size 0.49 mbFinding the formula of CuO teacher notes
Handout | PDF, Size 0.24 mbFind the formula of CuO scaffolded student sheet
Editable handout | Word, Size 0.5 mbFind the formula of CuO unscaffolded student sheet
Editable handout | Word, Size 0.51 mbFinding the formula of CuO slides
Presentation | PowerPoint, Size 0.6 mbFinding the formula of CuO teacher notes
Editable handout | Word, Size 0.47 mb
Additional information
This is a resource from the Practical Chemistry project, developed by the Nuffield Foundation and the Royal Society of Chemistry. The supporting resources were updated in 2026 by Harry Lord.
Practical Chemistry activities accompany Practical Physics and Practical Biology.
© Nuffield Foundation and the Royal Society of Chemistry






























2 readers' comments