Some metals are more reactive than others. Learners will investigate competition reactions of metals and determine a reactivity series of the four metals used
-

Download this
Download the scaffolded and unscaffolded student worksheets and teacher guidance (including answers) as MS Word and pdf and the presentation slides as MS Powerpoint and pdf.
Discover more resources from the Nuffield practical collection
In this experiment, a strip of metal is added to a solution of a compound of another metal. A more reactive metal will displace (push out) a less reactive metal from its compound. There are many ways of carrying out this series of reactions. The one described here uses a spotting tile, but you can adapt the same procedure for use with test tubes. The spotting tile method has several advantages:
- Very small quantities of chemicals are used.
- The whole set of experiments is displayed together, making comparison easier.
- Clearing up afterwards is simple and avoids metal deposits being left in sinks.
Video support and linked resources
A version of this experiment is included in our Reactivity of metals video (from 10:35), along with supporting resources, including illustrated technician notes, integrated instructions, worksheets, a structure strip and more.
Give careful thought to the distribution of the chemicals to the class. Distribute the solutions in test tubes, or in small bottles fitted with droppers for sharing between several pairs of learners. Issue the metals in sets but keep control of the magnesium ribbon, dispensing short lengths when required.
Make sure there are no flames alight so that learners are not tempted to burn pieces of magnesium and be alert to the possibility of pieces of magnesium being removed from the laboratory.
The experiment will take about 30 minutes.
Learning objectives
- Safely carry out microscale displacement reactions.
- Make inferences about reactivity from displacement reactions.
- Represent and explain displacement reactions using equations and particle diagrams.
- Plan a practical to allow further inferences to be made from data.
The practical allows learners to safely carry out microscale displacement reactions and make inferences about reactivity (LO1 and 2).
Completion of the follow-up questions will allow learners to succeed in LO3 and LO4.
Please note: if learners are unfamiliar with the word ‘inferences’ used in several of the learning objectives, explain that it is a conclusion reached using evidence collected from practical and scientific knowledge.
Scaffolding
There are two version of the student worksheet: scaffolded (✪) and unscaffolded (✪✪). The scaffolded sheet offers more support to allow learners to access the questions. For example, learners choose from a series of prepopulated answers or fill in gaps for longer answer questions.
Equipment
Apparatus
- Safety glasses
- Spotting tile, with at least 16 depressions (or two smaller tiles)
- Dropping (teat) pipette
- Beaker (100 cm3)
- Felt tip pen or other means of labelling
Chemicals
Access to about 5 cm3 each of the following 0.1 M metal salt solutions:
- Copper(II) sulfate (or nitrate(V))
- Magnesium nitrate(V)
- Iron(III) nitrate
- Zinc chloride (or nitrate(V))
Five samples, approximately 1 cm lengths or squares, of each of the following metals:
- Copper turnings
- Magnesium ribbon
- Iron filings (or small nails)
- Zinc (powder or small granules)
Chemical notes
- Dispense solutions in 5 cm3 beakers to each pair of learners or in small bottles fitted with droppers to groups of learners.
- Distribute metals in approximately 1 cm lengths or squares of ribbon or foil, cleaned with emery paper and as similar in size as possible.
Health, safety and technical notes
- Read our standard health and safety guidance and carry out a risk assessment before running any live practical.
- Wear safety glasses throughout.
- Beware of sharp edges when manipulating the metal strips.
- Copper(II) sulfate solution, CuSO4(aq), causes eye damage and is TOXIC to aquatic life. See CLEAPSS Hazcard HC027c, refer to SSERC or contact your local safety advisory body.
- Magnesium ribbon, Mg(s), is FLAMMABLE and gives off highly flammable gases in contact with acids. Do NOT leave in a place where pupils would have potentially unsupervised access. See CLEAPSS Hazcard HC059a, refer to SSERC or contact your local safety advisory board.
- Zinc powder, , is FLAMMABLE and hazardous to the aquatic environment. See CLEAPSS Hazcard HC107, refer to SSERC or contact your local safety advisory body.
- The following chemicals and substances are of low hazard:
For alternative guidance, refer to SSERC or contact your local advisory board.
Procedure
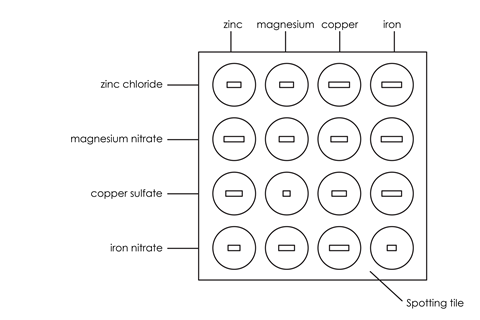
- Using a dropping pipette, put a little of the zinc chloride (or nitrate) solution in four of the depressions in the spotting tile, using the illustration below as a guide (or alternatively use the microscale integrated instruction sheet in the presentation slides). Label this row with the name of the solution. Rinse the pipette well with water afterwards.
- Do this for each metal ion solution in turn, rinsing the pipette when you change solution.
- Put a piece of each metal in each of the solutions, using the illustration as a guide.
- Over the next few minutes observe which mixtures have reacted and which have not.
Teaching notes
Remind the class that they are looking for cases where one metal displaces another. Some of the solutions are slightly acidic, so bubbles of hydrogen are sometimes seen. Explain that this does not count as displacement of one metal by another.
Get the class to tell you what they think the order of reactivity is, while they still have the evidence in front of them, so that apparent discrepancies can be resolved.
Downloads
Displacement reactions between metals and their salts lesson slides
Handout | PDF, Size 2.14 mbDisplacement reactions between metals and their salts student sheet
Handout | PDF, Size 0.27 mbDisplacement reactions between metals and their salts scaffolded student sheet
Handout | PDF, Size 0.31 mbDisplacement reactions between metals and their salts teacher notes
Handout | PDF, Size 0.38 mbDisplacement reactions between metals and their salts lesson slides
Editable handout | PowerPoint, Size 25.75 mbDisplacement reactions between metals and their salts student sheet
Editable handout | Word, Size 0.51 mbDisplacement reactions between metals and their salts scaffolded student sheet
Editable handout | Word, Size 0.54 mbDisplacement reactions between metals and their salts teacher notes
Editable handout | Word, Size 0.52 mb
Additional information
This is a resource from the Practical Chemistry project, developed by the Nuffield Foundation and the Royal Society of Chemistry. This collection of over 200 practical activities demonstrates a wide range of chemical concepts and processes. Each activity contains comprehensive information for teachers and technicians, including full technical notes and step-by-step procedures. Practical Chemistry activities accompany Practical Physics and Practical Biology.
The experiment is also part of the Royal Society of Chemistry’s Continuing Professional Development course: Chemistry for non-specialists.
© Nuffield Foundation and the Royal Society of Chemistry


















No comments yet