Create a small explosion inside a tin can using cornflour in this teacher demonstration
In this experiment, students observe what happens when cornflour is sprayed into the flame of a candle burning inside a large tin can with the lid on. The resulting small explosion caused by rapid combustion of the cornflour blows the lid off the tin.
The reaction dramatically illustrates the conversion of the chemical energy stored in foodstuffs into heat and other forms of energy, and can also be used to show the effect of surface area on the rate of chemical reaction.
This is a brief demonstration, taking about five minutes – but of course may have to be repeated on demand.
Equipment
Apparatus
- Safety screen
- Eye protection for teacher
- Large coffee tin, 500 g catering size, with metal lid (see notes 4 and 5 below)
- Glass funnel, small, 2–3 cm diameter
- Rubber bung, 13 mm, one hole
- Candle, short piece, or nightlight
- Pipette filler, rubber bulb, about 50–100 cm3
- Rubber tubing, short lengths, to join pipette filler to funnel stem
- Wooden splint and matches to light candle
Chemicals
- Cornflour, dry, a few grams required for each demonstration
Health, safety and technical notes
- Read our standard health and safety guidance.
- Wear eye protection. The experiment must be conducted behind firmly fixed safety screens close to the can, which protect both class and teacher. The class should be seated sufficiently far back from the demonstration to avoid any risk of being hit by the flying tin lid.
- Cornflour – as an alternative to cornflour, other similar, oxidisable, fine powders can be used, eg custard powder, icing sugar or lycopodium powder. (Note: lycopodium powder is a form of pollen, which may cause sensitisation or hay fever-like symptoms in susceptible individuals. If the demonstration works well, very little lycopdium powder should enter the air in the room.) The powder usually needs to be dried in an oven at about 80 °C.
- The apparatus needs to be assembled as shown in the diagram below before the lesson. Make a neat hole in the side of the coffee tin, near the base, of suitable size to take the one-holed rubber bung. Insert the funnel into the hole in the bung so that the stem protrudes outwards from the narrow end of the bung. Fit the bung carrying the funnel into the hole from inside the can, so that it is pointing slightly upwards - see diagram. Connect the pipette filler bulb to the funnel stem using short lengths of glass and rubber tubing. Place a short length of candle (or a nightlight) inside the coffee tin and stick it down with a little molten wax.
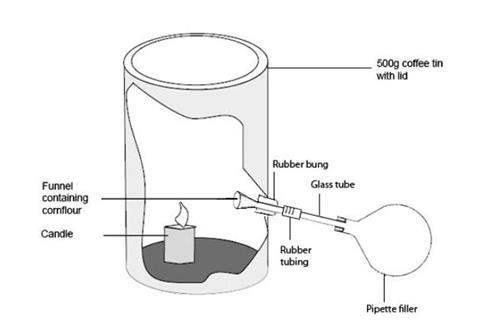
- This apparatus can be improvised in a number of ways, but it is important that the resulting apparatus can blow a cloud of cornflour into the candle flame when the pipette filler is squeezed. An alternative arrangement is to place a small crucible inside the can, to contain the cornflour, and replace the funnel with a glass tube bent at right-angles, so that it’s tip is directed downwards into the crucible. Some prior experimentation and adjustment may be needed to ensure the demonstration works well.
Procedure
- Place a few grams of cornflour in the funnel. This may be more easily done before finally putting the funnel in place inside the can, and connecting it to the pipette bulb.
- Light the candle and quickly fit the tin lid. Quickly (before the candle goes out) give the pipette filler a rapid squeeze to blow the fine cornflour powder into the candle flame, keeping your head well back. The resulting rapid combustion of the finely-divided cornflour blows the lid off the tin.
Teaching notes
The demonstration can be set in context with stories about the explosion risks of powders such as sawdust from sanders, custard powder and flour in mills and coal dust explosions in mines. The latter can be contrasted with the difficulty often encountered in igniting large lumps of coal when lighting a fire.
The increase in surface area caused by fine sub-division can be illustrated with eight identical wooden cubes (eg Tillich’s bricks) built into a 2 x 2 x 2 cube. The surface area of this can be shown to be (2 x 2) x 6 sides = 24 units. When the cube is broken into its eight constituent bricks, the total surface area is now (1 x 1) x 6 sides x 8 cubes = 48 units. Also, shading the outside of the 2 x 2 x 2 cube with chalk before breaking it up will show that more surface is exposed when the cube is broken up because each of the smaller cubes will have three shaded and three unshaded faces.
Cornflour (a carbohydrate) burns rapidly because of its high total surface area to volume ratio, which allows oxygen in the air to come into to contact with the fuel easily. This demonstration is a good illustration of energy changes in chemical reactions, and that some compounds have a lot of energy ‘locked up’ in them. Students can be asked to identify the types of energy involved as the lid flies off - heat, light, sound, kinetic and potential (the lid at the top of its trajectory). The idea of activation energy could be introduced to a suitable class.
Warning
There are many video clips available online which demonstrate such powder explosions, including some from school laboratories. In many of these, safety was clearly not a prime consideration for those performing the experiments. These should not be shown in school in case they serve to encourage very hazardous experimentation by children, possibly culminating in injury or worse.
Additional information
This is a resource from the Practical Chemistry project, developed by the Nuffield Foundation and the Royal Society of Chemistry.
Practical Chemistry activities accompany Practical Physics and Practical Biology.
© Nuffield Foundation and the Royal Society of Chemistry


















1 Reader's comment