Download this
The technician notes as MS Word or pdf and accompanying slides as MS Powerpoint or pdf
Watch this
Watch the video and download the technician notes from the Education in Chemistry website: rsc.li/3oPsEmV
An appealing demonstration that delights the eyes, ears, and stimulates (!) the nose involves the silicon analogue of the alkanes: the silanes. The creation and pyrophoric reaction of these compounds in air is an excellent way to explore a range of chemistry topics including redox and acid-base reactions, combustion, kinetics, and structure and bonding in macromolecular as well as simple covalent substances. It has even been chosen as context for GCSE exam questions.
Kit
- 2 g magnesium powder (flammable)
- 1 g silica/silver sand/quartz sand
- 50 cm3 2 M hydrochloric acid
- Medium borosilicate test tube (approx 15 x 180 mm)
- Clamp and stand
- Bunsen burner
- Heat-resistant mats
- 250 cm3 beaker
- Spatula
- Access to oven and desiccator
Preparation
Weigh out 1 g of silica and 2 g of magnesium powder. Thoroughly dry these and the test tube for a few hours in an oven at approx 80°C, then leave to cool in a desiccator until immediately before the demonstration.
Use heat-resistant mats to protect the bench. Clamp the tube with the open end angled slightly above the horizontal. To achieve homogeneity, mix the powders thoroughly by pouring them from one scrap of folded paper to another at least 10 times. Spread the mixture along the lower third of the tube, ensuring there is clearance above the powder. Pour 50 cm3 of 2 M hydrochloric acid into a 250 cm3 beaker.
Set up safety screens to protect the audience and demonstrator.
In front of the class
The class should be positioned two metres away wearing eye protection. Wear splash-proof goggles and hold a lit Bunsen with a roaring flame to the end of the powder furthest from the open end of the tube. After a few moments of strong heating, the glass will darken and may deform slightly and the mixture will begin to glow bright orange. Move the Bunsen along the tube to chase the glow along the powder until all of it has reacted. Be prepared for a pop of hydrogen if the tube or powders were not dried sufficiently.
Ensure the room is well ventilated for the next part of the demonstration as the gas has a pungent smell. Once the tube has completely cooled, scrape the sides of the tube to dislodge the products. Use caution, as the weakened glass may break. Pour the powder into the waiting beaker of acid. As it sinks to the bottom, it will begin to produce bubbles of gas. These will rise to the surface and spontaneously combust with pops and crackles on contact with the air.
Health and safety and disposal
- CLEAPSS members should consult PX059 and HC059A.
- Wear splash-proof goggles and use safety screens to protect both the audience and demonstrator.
- Students should remain two metres away wearing eye protection.
- Ensure powders are thoroughly dried before starting the reaction.
- Leave the tube in submerged water to thoroughly remove any remaining silicides and then dispose of in the glass waste.
Teaching goal
Industrially, these types of reactions are of great importance as silanes and halosilanes are used in the production of high purity silicon for semiconductor applications through the use of chemical vapour deposition (CVD).
The first displacement reaction demonstrates silicon’s relative position on the activity series compared with magnesium, as well as the high melting point of silica, a macromolecular substance. Silicon can go on to further react with magnesium.
Equation 1: 2Mg(s) + SiO2(s) → 2MgO(s) + Si(s)
Equation 2: Si(s) + 2Mg(s) → Mg2Si(s)
On initial addition of the product mixture to the acid, the metal oxide reacts and dissolves away to reveal the silicon following a classic acid–base equation. The silicon/silicide is now also able to react with the acid to produce a range of small hydrides analogous to the alkanes with the general formula Sin H2n+2.
Equation 3: MgO(s) + 2HCl(aq) → MgCl2(aq) + H2O(l)
Equation 4: Mg2Si(aq) + 4HCl(aq) → SiH4(g) + 2MgCl2(aq)
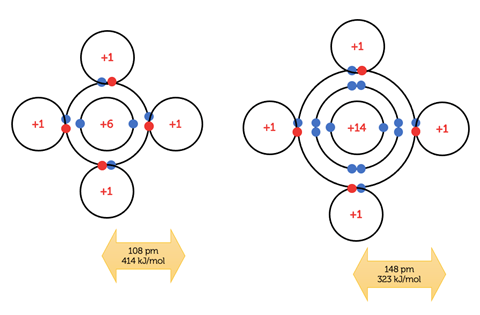
These small silanes float to the surface, where combustion follows the same initial pathway as that of methane, allowing for a meaningful comparison of activation energies to be made in association with bond enthalpy values. The longer, weaker, Si–H bonds will break more easily, leading to more frequent successful collisions at a lower temperature than in the methane–air mixture.
Downloads
Pyrophoric silanes – technician notes
Experiment | Word, Size 69.48 kbPyrophoric silanes – technician notes
Experiment | PDF, Size 42.27 kbChasing the glow - slides
Presentation | PowerPoint, Size 2 mbChasing the glow - slides
Presentation | PDF, Size 1.95 mb






















No comments yet