Try this class practical or demonstration to illustrate the displacement of copper from copper(II) sulfate using aluminium foil
In this experiment, students add aluminium cooking foil to copper(II) sulfate solution and observe no reaction. They then add and dissolve sodium chloride, producing a vigorous displacement reaction which illustrates the reactivity of aluminium. The solution gets very hot, the aluminium dissolves and red copper becomes visible.
The class practical can take about 30 minutes to complete. A flexicam would work well if this is to be done as a demonstration and allow students a clearer view of what is going on.
Equipment
Apparatus
- Eye protection (goggles)
- Conical flask, 100 cm3
Chemicals
- Aluminium foil, 2 cm x 2 cm
- Copper(II) sulfate solution, 0.8 M (HARMFUL), 20 cm3
- Sodium chloride, 2–3 g
Health, safety and technical notes
- Read our standard health and safety guidance.
- Wear eye protection (goggles) throughout and disposable nitrile gloves.
- Aluminium foil, Al(s) – see CLEAPSS Hazcard HC001A.
- Copper(II) sulfate solution, CuSO4(aq), 0.8 M (HARMFUL, DANGEROUS FOR THE ENVIRONMENT) – see CLEAPSS Hazcard HC027c and CLEAPSS Recipe Book RB031.
- Sodium chloride, NaCl(s), (table salt) – see CLEAPSS Hazcard HC047b.
- Ensure the aluminium foil is completely consumed by the reaction before disposal to prevent a continued exothermic reaction in the rubbish bin. Use plenty of copper(II) sulfate solution and sodium chloride to ensure a complete reaction.
Procedure
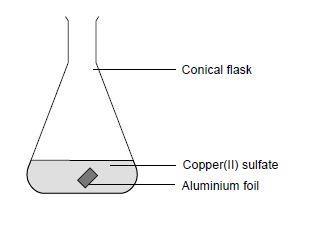
- Measure approximately 20 cm3of copper(II) sulfate solution into the conical flask.
- Add a square of aluminium foil.
- Look for signs of a reaction.
- Add a spatula of sodium chloride and stir to dissolve.
- Observe any changes. If nothing happens, add more sodium chloride. Has displacement of copper from copper(II) sulfate occurred?
Student questions and example table
- Before the sodium chloride is added, does any reaction occur?
- After adding sodium chloride, does the aluminium appear more or less reactive?
- How does the addition of sodium chloride affect this change?
- Write ‘yes’ or ‘no’ to fill in the table below.
| Observations | Before the sodium chloride is added | After the sodium chloride is added |
|---|---|---|
| Bubbles observed | ||
| Colour changes | ||
| Temperature change | ||
| Copper observed |
Teaching notes
Aluminium does not show its true reactivity until the oxide layer is disturbed. Sodum chloride disturbs this oxide layer. Scratches on the surface of the oxide layer allow chloride ions to react with aluminium, this effects the cohesiveness of the oxide layer. This allows reaction with the copper(II) sulfate. Remind students what copper looks like, so that they know what they are looking for.
Answers to student questions
- Aluminium appears less reactive than copper. The aluminium foil appears unable to displace copper from copper(II) sulfate solution.
- Now aluminium is more reactive because it displaces copper. Aluminium + copper(II) sulfate → copper + aluminium sulfate
- Scratches on the surface of the oxide layer allow chloride ions to react with aluminium, this effects the cohesiveness of the oxide layer. This allows a simple exchange reaction with the copper(II) sulfate. The protective oxide layer forms instantly the aluminium is exposed to the air.
Additional information
This is a resource from the Practical Chemistry project, developed by the Nuffield Foundation and the Royal Society of Chemistry.
Practical Chemistry activities accompany Practical Physics and Practical Biology.
© Nuffield Foundation and the Royal Society of Chemistry

















4 readers' comments