Use this demonstration to illustrate the conditions required to start combustion, and how to put out a pan fire safely
In this experiment, students observe as cooking oil in a crucible catches fire over a Bunsen burner, simulating a fat-pan or chip-pan catching fire in a kitchen. They then watch as the fire is extinguished by placing a ‘tray’ over the top of the crucible, reducing the air available to the fire.
For school use, this must only be done as a demonstration. The demonstrator must have practiced the demonstration beforehand until confident that the procedure can be done safely in front of a class, when it should take 10–15 minutes.
Equipment
Apparatus
- Face shield for teacher
- Eye protection for class
- Heat resistant gloves
- Crucible, 25 mm diameter, nickel or steel (see note 3 below)
- Pipeclay triangle to support crucible (see note 3)
- Tripod
- Bunsen burner
- Heat resistant mats (see note 4)
- Safety screens (see note 5)
- A small square of hardboard or aluminium
- Test tube, fixed securely to end of 1 metre pole (see note 6)
Chemicals
- Cooking oil, 3 cm3 per demonstration
Health, safety and technical notes
- Read our standard health and safety guidance.
- Wear eye protection throughout. Teacher to wear face shield and heat resistant gloves. Class to wear eye protection and must be kept not less than four metres back. Safety screens must be positioned and secured to protect both students and the demonstrator. The experiment must not be sited below a light fitting.
- Wedge the nickel crucible firmly and upright in the pipeclay triangle on the tripod. It must not tip over when the flame is smothered by the demonstrator. The wires of the triangle may need to be bent over the tripod.
- Protect the demonstration bench from hot burning fat by covering with an array of heat resistant mats.
- Arrange safety screens and secure in place so they protect both demonstrator and the class.
- The test tube should be held firmly, eg in a test tube holder, and strapped to the end of a long pole (a metre rule will suffice) so that the tube will not fall off when engulfed in flame during the demonstration.
Procedure
- Place about 5 cm3 of water in the test tube ready for use during the demonstration.
- Pour 3 cm3 of cooking oil into the crucible and place a lighted Bunsen burner beneath it.
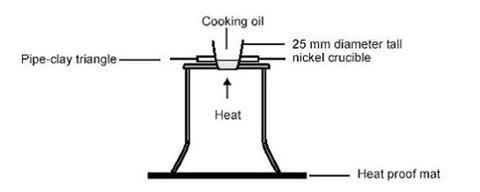
- Once the oil catches fire, switch off the gas supply to the Bunsen burner and extinguish the flame by placing a small square of hardboard or aluminium over the crucible to simulate placing a tray over a burning chip pan to remove the oxygen from the fire.
- Explain that a damp tea-towel would also extinguish the fat-pan fire. In this demonstration this method is unsuitable, as it could knock the apparatus off the tripod, but for a real fat-pan fire it is a good method.
- Remove the square and light the Bunsen burner again until the cooking oil re-ignites.
- Switch off the gas supply to the Bunsen burner, and holding the pole with the test tube containing water attached at arm’s length, add the water to the burning oil. This will cause a ball of fire to rise about a metre, effectively demonstrating the hazard of attempting to put out a fat-pan fire with water.
Teaching notes
The procedure described is hazardous and risks are made acceptable only by adherence to the restrictions and precautions advocated. In addition to the precautions described above:
- The demonstration must NOT be done in a fume cupboard.
- The quantities prescribed must NOT be exceeded; do NOT be tempted to use more cooking oil.
- A squat crucible must NOT be used as it ejects the hot fat sideways.
- A porcelain crucible is NOT safe as it is liable to break.
This demonstration can be linked to the teaching of the ‘fire triangle’ as well as to the more general aspects of assessing risks and taking action to reduce risks to themselves and others. Chip-pan fires cause one-fifth of all accidental dwelling fires in the UK each year. As well as the damage they can cause to people’s homes, these fires also injure around 4,000 people every year. (Reproduced from CLEAPSS Guide, L195: ’Safer Chemicals, Safer Reactions’, section 9.5 by permission of CLEAPSS®.)
Further information
This kitchen fire safety document distributed by the West Sussex fire and rescure service provides guidance on how to prevent fires, cook safely and respond to a pan fire.
Downloads
Kitchen fire safety
PDF, Size 0.49 mb
Additional information
This is a resource from the Practical Chemistry project, developed by the Nuffield Foundation and the Royal Society of Chemistry. This collection of over 200 practical activities demonstrates a wide range of chemical concepts and processes. Each activity contains comprehensive information for teachers and technicians, including full technical notes and step-by-step procedures. Practical Chemistry activities accompany Practical Physics and Practical Biology.
The experiment is also part of the Royal Society of Chemistry’s Continuing Professional Development course: Chemistry for non-specialists.
© Nuffield Foundation and the Royal Society of Chemistry

















No comments yet