Use this class practical to determine the relative atomic mass of magnesium using its reaction with hydrochloric acid
In this experiment, learners react magnesium ribbon with dilute hydrochloric acid to produce hydrogen gas. They then use the measured volume of hydrogen gas produced and the mass of magnesium to calculate the mass of magnesium required to produce one mole of hydrogen molecules. From this, learners can deduce the relative atomic mass of magnesium.
-

Download this
A ready-to-go practical lesson with classroom slides, scaffolded and unscaffolded student worksheets and teacher guidance, including full technical notes and answers to all questions.
Discover more resources from the Nuffield practical collection
This is a class experiment suitable for learners who already have a reasonable understanding of the mole concept, and are beginning to use chemical equations to perform calculations.
A total of 45 minutes should be adequate for the class to obtain and record their results, including the time taken by the teacher to demonstrate the procedure, and allowing an average of five minutes for each learner to measure the mass of their magnesium ribbon. However, the timing depends on the number of top-pan balances available and the skill with which learners can use the balance to sufficient accuracy.
Learning objectives
- Measure the volume of hydrogen gas produced during an acid-metal reaction.
- Record accurate measurements of mass and volume in a results table.
- Determine a molar ratio during a chemical reaction using balanced chemical equations.
- Estimate the relative atomic mass of magnesium using your experimental results.
Success criteria
Learners will first carry out the practical to measure the volume of hydrogen gas produced (LO1, LO2). Using molar volume calculations, they will determine the moles of hydrogen produced. By linking this to the balanced chemical equation, learners will therefore deduce the moles of magnesium required (LO3) and hence the relative atomic mass (using the starting mass recorded)(LO4).
Scaffolding
Two versions of the worksheet are available: scaffolded and unscaffolded. The scaffolded sheet offers more support to allow learners to access the questions. The answers to the worksheets are available in the teacher notes.
The follow-up calculation uses two different equations and you can choose to work through this collaboratively with your class using the lesson slides to support understanding. The equations are given in the scaffolded sheet.
Technician notes
Read our standard health and safety guidance and carry out a risk assessment before running any live practical.
Equipment
Apparatus
- Safety glasses
- Fine emery paper, a few cm, x2
- Burette, 50 cm3 (see note 1 below)
- Burette stand
- Funnel, small
- Beaker, 100 cm3
- Beaker, 250 cm3
- Access to a top-pan balance, accurate to +/– 0.001 g (see note 2 and 3 below)
- Access to room temperature and pressure measurements (see note 4 below)
Apparatus notes
- Make sure the burette taps are free from leakage, operate smoothly and are secure in their sockets.
- A balance reading to only +/– 0.01 g does not have sufficient accuracy for the procedure used in this experiment, where the maximum possible volume of hydrogen that can be collected is only 0.002 mol, which would be produced by 0.048 g of magnesium.
- If a balance accurate to 0.001 g is not available, you can obtain reasonable results by measuring the mass of a much longer (eg 30 cm) piece of magnesium ribbon beforehand on a balance with an accuracy of 0.01 g, measuring its length and then cutting it accurately into 3 cm lengths. Using the mass and length of the long piece of magnesium, the average mass of a 3 cm length can be calculated with sufficient accuracy.
- For the additional challenge, if a barometer is not available in the laboratory, obtain an up-to-date reading of atmospheric pressure shortly before the lesson, eg from a local weather website. You will also need to measure room temperature.
Chemicals
- Hydrochloric acid, 2 M, (IRRITANT), 25 cm3
- Magnesium ribbon (FLAMMABLE), approximately 3–4 cm length
Safety and hazards
- Wear eye protection throughout.
- Dilute hydrochloric acid, HCl(aq), (IRRITANT at concentration used)
- see CLEAPSS Hazcard HC047a and CLEAPSS Recipe Book RB043.
- Provide the hydrochloric acid in small bottles or corked conical flasks, labelled, suitable for pouring the acid into the burette.
- Magnesium ribbon, Mg(s)
- see CLEAPSS Hazcard HC059A.
- Clean the magnesium ribbon with emery paper to remove the grey oxide layer, so that it appears shiny and metallic. Cut the ribbon into lengths of 3–4 cm (which will yield around 30 cm3 of hydrogen when reacted).
- Take steps to prevent theft of magnesium ribbon. Reels of magnesium ribbon should not be left out in the laboratory. It is good practice to have a limited number of pre-cut lengths and to hand these out to learners as needed.
- Magnesium chloride solution, MgCl2(aq), is currently not classified as hazardous.
Disposal
- Dispose of hydrochloric acid by diluting in water to below 0.1M. Pour the diluted solution down a foul-water drain with further dilution.
- Dispose of small amounts of magnesium by adding slowly to 1 M ethanoic acid solution. Test the solution with indicator and add more ethanoic acid until the mixture is just acidic. The reaction generates a lot of heat. Pour the neutralised mixture down a foul-water drain with further dilution. NEVER dispose of flammable chemicals in the normal refuse.
- Dispose of the product, magnesium chloride, by pouring the solution down a foul-water drain.
Method
- Accurately measure the mass, to the nearest 0.001 g, of a length of magnesium ribbon, approximately 3–4 cm long. The mass should lie between 0.020 g and 0.040 g.
- Make sure the burette tap is closed. Use a small funnel to pour 25 cm3 of dilute hydrochloric acid into the burette, followed carefully by 25 cm3 of water. Try to avoid mixing of the two liquids as far as possible. Accurate volume measurements are not needed. This should leave a space of at least 5 cm between the liquid and the top of the burette.
- Carefully push the magnesium ribbon into the open end of the burette, pushing the strip in the middle so that the springiness of the strip holds it in place against the glass. Do not allow it to contact the liquid at this stage.
- Add about 50 cm3 of water to a 250 cm3 beaker.
- Rest the top of the burette gently on the lip of the beaker, then quickly turn the burette upside-down and lower the end beneath the water in the beaker. If this is done quickly and carefully (the teacher may wish to demonstrate this first), little or no liquid will be lost. Clamp the burette vertically in this upside-down position.
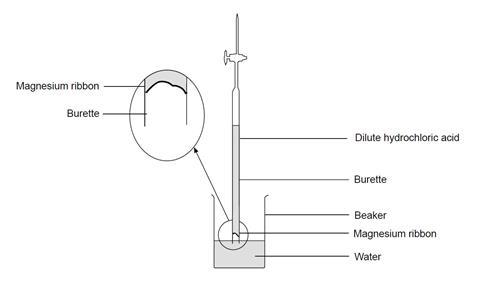
- Without delay, check that the liquid level in the burette is on the scale – if it is not, open the tap momentarily to allow the level to drop on to the scale.
- Take the burette reading of the liquid level. (Note: the scale is also now upside-down!)
- As the acid diffuses downwards, the magnesium begins to react. Allow the metal to react completely.
- Once the liquid level has ceased to change, and no more gas bubbles are being formed, take the final burette reading, and record the result.
Teaching notes
Demonstrate the procedure beforehand. The inversion is not difficult, and it is not necessary to put a finger over the open end. Warn learners not to fold the magnesium ribbon, but to push it into the burette so that it is retained under its own tension.
You can collect results from the class on a spreadsheet to enable a discussion about their reliability.
Learners can perform the resulting calculation at various levels. They need to be able to understand the equation:
Mg + 2HCl → MgCl2 + H2
and to use it to recognise that one mole of magnesium will yield one mole of hydrogen molecules. From the results, the least required of learners would be to perform a proportionality calculation to determine the mass of magnesium that would have yielded 24,000 cm3 of hydrogen.
As an additional challenge, show some learners the ideal gas equation and ask them to convert the volume of gas collected under known conditions in the experiment to standard temperature and pressure, then determine the mass of magnesium that would have yielded 24,000 cm3 of hydrogen.
Follow-up questions
Answers to the follow-up questions in the student worksheets and on the lesson slides can be found in the teacher notes.
Further information
Most websites deal with instrumental procedures for determining atomic masses, such as mass spectrometry. Many of these are concerned with levels of detail far beyond what is appropriate even for more advanced students. However, Chemguide offers a fairly detailed treatment of mass spectrometry for determining atomic masses suitable for advanced students.
Downloads
Determining the relative atomic mass of magnesium unscaffolded student sheet
Handout | PDF, Size 0.33 mbDetermining the relative atomic mass of magnesium scaffolded student sheet
Handout | PDF, Size 0.34 mbDetermining the relative atomic mass of magnesium teacher notes and answers
Handout | PDF, Size 0.26 mbDetermining the relative atomic mass of magnesium lesson presentation slides
Presentation | PDF, Size 0.97 mbDetermining the relative atomic mass of magnesium unscaffolded student sheet
Editable handout | Word, Size 0.59 mbDetermining the relative atomic mass of magnesium scaffolded student sheet
Editable handout | Word, Size 0.58 mbDetermining the relative atomic mass of magnesium teacher notes and answers
Editable handout | Word, Size 0.45 mbDetermining the relative atomic mass of magnesium lesson presentation slides
Presentation | PowerPoint, Size 10.29 mb
Additional information
This is a resource from the Practical Chemistry project, developed by the Nuffield Foundation and the Royal Society of Chemistry. This collection of over 200 practical activities demonstrates a wide range of chemical concepts and processes. Each activity contains comprehensive information for teachers and technicians, including full technical notes and step-by-step procedures. Practical Chemistry activities accompany Practical Physics and Practical Biology.
The supporting resources were updated in 2026 by Louise Glynn.
© Nuffield Foundation and the Royal Society of Chemistry
























2 readers' comments