In this demonstration, students observe the highly exothermic reaction between aluminium and iron(III) oxide that produces molten iron
The reaction is violent but safe provided the procedures are followed exactly. Some teachers have had accidents when performing the procedure outside in a strong breeze; the powders blew into the flame, caught fire and caused burns to the hand and/or face. Siting the demonstration in a fume cupboard has caused damage to the cupboard.
The method described here is performed on a laboratory bench and produces limited fumes. Do NOT do this demonstration in a fume cupboard or outdoors. It produces a result within seconds of setting it off because the water cools the iron down very quickly. A rehearsal is essential if this experiment has not been done before.
There have been occasional reported explosions when using methods similar to this. It is essential not to exceed the stated quantities and that the demonstrator and students are protected by safety screens.
The bench should be clear of combustible materials and protected with a sheet of hardboard or heat-resistant mats. The demonstrator must have room to move quickly away to a safe distance.
The demonstration takes about 10 minutes to carry out if the apparatus is set up and the solid reagents are weighed in advance.
Equipment
Apparatus
- Eye protection (note 2)
- Safety screens (note 3)
- Filter paper, 11 cm diameter
- Pipeclay triangle
- Tripod
- Plastic beaker or thick-walled glass beaker, 1 dm3 (must fit between tripod legs)
- Sand (see diagram)
- Heat-resistant mats
- Plastic magnetic retriever or small bar magnet
Chemicals
For the thermite mixture (note 6):
- Aluminium powder (medium grade) (HIGHLY FLAMMABLE), 3 g
- Iron(III) oxide, 9 g
For the igniter (notes 9 and 10):
- Domestic sparkler, 16 cm long (sold for indoor use)
Health, safety and technical notes
- Read our standard health and safety guidance.
- Both the demonstrator and all observers must wear eye protection. For the demonstrator this must be goggles or a face shield.
- Safety screens must be used to surround the apparatus. In addition to wearing eye protection, students should stand further than 4 m from the reaction.
- The demonstrator should wear a laboratory coat (the experiment can become messy at the end).
- The thermite reaction can trigger heat or smoke sensors; you cannot do the experiment in a laboratory fitted with smoke sensors.
- It is important that the iron(III) oxide used in this demonstration is absolutely dry. An hour or so in a warm oven, or heating in an evaporating dish over a Bunsen flame, should suffice. The oxide should be allowed to cool completely before mixing. The weighed quantities of iron(III) oxide (9 g maximum) and aluminium powder (3 g maximum) may be thoroughly mixed beforehand by repeatedly pouring the mixture to and fro between two pieces of scrap paper (never stir with a metal spatula), and then stored for the demonstration in a suitable container labelled ‘Thermite mixture’.
- Aluminium powder, Al(s), (HIGHLY FLAMMABLE) – see CLEAPSS Hazcard HC001A.
- Iron(III) oxide, Fe2O3(s) – see CLEAPSS Hazcard HC055A.
- The Igniter for the reaction is a domestic sparkler. It should be one for indoor use but need not be handheld. The sparkler needs to be longer than 10 cm to ensure there is time to retreat to a safe distance. A 16 cm sparkler length is ideal and the handle should be cut off using tin snips or pliers. Sparklers longer than 16 cm may become top-heavy. Avoid cutting into excess explosive; this causes it to crack, and it can fall off dangerously before the demonstrator can retire to a safe distance.
- Due to the UK Explosives Regulations 2014 alternative igniter mixtures (such as magnesium powder, barium nitrate and magnesium ribbon) on scales larger than 0.5 g combined weight cannot be used in schools without an Explosives Certificate issued by the police. Igniter mixtures at or below 0.5 g result in a less reliable ignition, so a sparkler should be used.
The demonstrator may wish (or be persuaded by the audience) to do a repeat demonstration. In this event it is important to keep the second set of materials well away from the first demonstration site. - Disposal: if the sparkler fails to set off the thermite mixture, dispose of the unreacted mixture by pouring it into dilute (2 M) hydrochloric (1200 cm3) or sulfuric acid (600 cm3) in a beaker in a fume cupboard and leave to stand overnight. You can use 1 M dilute acid but will need to double the volumes of acid used. Filter off the solids and then place in the non-recycling waste and dilute down the liquid with lots of water.
Procedure
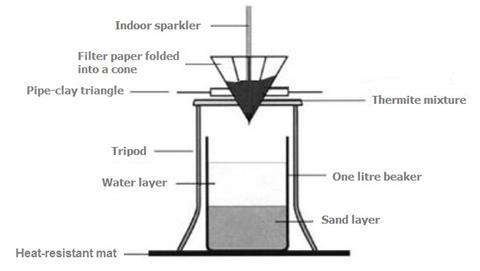
- Fold one 11 cm diameter filter paper into a cone shape.
- Into a 1 dm3 beaker, pour sand until it is two-fifths (2/5) full and then add water until it is four-fifths (4/5) full.
- Cover an area of the bench with several heat-resistant mats and place the beaker in the centre. Set up the equipment as shown in the diagram above and surround it with safety screens. Add the thermite mixture (see note 6) to the filter paper cone sitting in the pipe clay triangle. WARNING: push the filter paper cone into the triangle firmly, so there is no tendency for it to pop out.
- Insert a domestic sparkler upright into the thermite mixture. 3–4 cm should be buried below the mixture with the rest extended above the filter paper cone. Light the sparkler with a Bunsen burner flame and retreat to a safe distance behind the safety screens. A very vigorous reaction should follow, with some sparks flying upwards. The very hot residue containing molten iron will fall through into the water.
- Once the reaction has stopped, remove the beaker and decant the water into the sink. Retrieve the iron formed with a magnet. Wash the iron under running water.
Teaching notes
The reaction is: iron(III) oxide + aluminium → aluminium oxide + iron.
This shows that aluminium is above iron in the reactivity series.
Once underway, the reaction is highly exothermic, rapidly reaching temperatures as high as 2000 °C, well in excess of the melting point of iron (1535 °C).
The practical use of this reaction is to weld railway lines together and could be mentioned.
The ‘thermite’ mixture is stable until strong heating is applied, hence the need for the domestic sparkler to initiate the reaction. Some of the igniter mixtures commonly used in the past are now covered by the UK Explosives Regulations 2014. This means that no more than 0.5 g of such mixtures can be used unless the school has an Explosives Certificate issued by the police. The sparkler method is preferred because igniter mixtures on scale of 0.5g or below are less reliable.
If you choose to show students video clips of thermite reactions from the internet, take care to avoid those that are carried out on a scale and in a manner which is hazardous.
More resources
Inspire learners and discover more ways chemists are making a difference to our world with our video job profiles.
Additional information
This is a resource from the Practical Chemistry project, developed by the Nuffield Foundation and the Royal Society of Chemistry.
Practical Chemistry activities accompany Practical Physics and Practical Biology.
The experiment is also part of the Royal Society of Chemistry’s Continuing Professional Development course: Chemistry for non-specialists.
© Nuffield Foundation and the Royal Society of Chemistry
This resource has been reviewed and updated with advice from CLEAPSS in response to the UK Explosives Regulations 2014.

















1 Reader's comment