Try this class practical to make samples of glass using lead oxide, zinc oxide and boric acid
In this experiment, students heat a mixture of lead oxide, zinc oxide and boric acid strongly until it melts, forming molten glass. They can colour their glass samples by adding traces of various transition metal oxides, before allowing beads of glass to cool in order to examine them.
This class experiment demands careful manipulation of very hot apparatus by students. Teachers will need to be satisfied that a class is capable of doing so in a safe and orderly manner before using this experiment.
The experiment itself may take up to 60 minutes, given the need for careful handling and weighing of toxic and harmful metal oxides, careful heating of the crucible to a high temperature with stirring of the contents, and finally adding a trace of a transition metal oxide to the melt with continuous stirring.
Equipment
Apparatus
- Eye protection
- Access to top-pan balance (±0.1 g) (see note 1 below)
- Boiling tube, 150 x 25 mm, Pyrex or similar
- Rubber bung, to fit boiling tube
- Spatula
- Crucible, low (squat) form, approx 15–20 cm3 capacity, with lid (see note 2 below)
- Pipe clay triangle (see note 2)
- Crucible tongs
- Bunsen burner
- Tripod
- Heat resistant mat
- Plastic weighing boats
- Paper clips, large enough to form a long stirrer wire when straightened
Apparatus notes
- The smooth progress of this experiment depends on each group having easy access to a top-pan balance weighing to +/- 0.1 g. Each group has to make three weighings, which may well take up to five minutes, so enough balances will be needed for three groups to each balance. In addition there is the issue of spillage and dust from some of the toxic oxides. It is therefore probably better if the mixture is made up in bulk in a fume cupboard in a large, self-sealing plastic bag. The mixture could then be distributed in small plastic bags. If these cannot be provided, then some or all of the three chemicals required may have to be supplied in pre-weighed amounts. Use disposable plastic weighing boats, if available.
- Crucibles: ceramic crucibles are best but are liable to breakage in class use. Stainless steel crucibles, if available, are more durable, but should be absolutely clean to avoid contamination by any metal oxides from previous use. The crucibles should sit securely in, not just resting on, the clay pipe triangles, which should also be in good condition (not broken or bent out of shape) to avoid the risk of crucibles falling through or tipping over during the experiment. It is suggested that the crucibles should be dedicated to this experiment. After use, the crucibles used to make this glass should be immersed in 1 M dilute nitric(V) acid (CORROSIVE) for cleaning. Allow the glass to dissolve, and dilute to 1 litre before pouring down a foul-water drain. The crucibles must be thoroughly dried before reuse.
Chemicals
- Boric acid (boracic acid) about 5 g
- Lead(II) oxide (TOXIC, DANGEROUS FOR THE ENVIRONMENT), about 8 g
- Zinc oxide (DANGEROUS FOR THE ENVIRONMENT), about 1 g
- Chromium(III) oxide, trace
- Cobalt(II) oxide (HARMFUL), trace
- Copper(II) oxide (HARMFUL, DANGEROUS FOR THE ENVIRONMENT), trace
- Manganese(IV) oxide (HARMFUL), trace
Health, safety and technical notes
- Read our standard health and safety guidance.
- Wear eye protection throughout. Avoid inhaling lead oxide dust. Wash hands after handling lead compounds.
- This recipe for making a glass uses several toxic and harmful chemicals, so quantities for student access should be minimised as indicated above, and made available to each working group if possible in small snap-lid plastic sample pots or similar containers. Note also that some of the transition metal oxides used can be very expensive to buy, so it is worth keeping quantities small for this reason as well. Do not use nickel(II) oxide (TOXIC) in this experiment.
- Boric acid, H3 BO3 (s) – see CLEAPSS Hazcard HC014a.
- Lead(II) oxide, PbO(s), (TOXIC, DANGEROUS FOR THE ENVIRONMENT) – see CLEAPSS Hazcard HC056.
- Zinc oxide, ZnO(s), (DANGEROUS FOR THE ENVIRONMENT) – see CLEAPSS Hazcard HC108b.
- Chromium(III) oxide, Cr2 O3 (s) – see CLEAPSS Hazcard HC024. Note that chromium(III) oxide must not be confused with chromium(VI) oxide, which is a VERY TOXIC substance.
- Copper(II) oxide, CuO(S), (HARMFUL, DANGEROUS FOR THE ENVIRONMENT) – see CLEAPSS Hazcard HC026.
- Cobalt(II) oxide, CoO(s), (HARMFUL) – see CLEAPSS Hazcard HC025.
- Manganese(IV) oxide, MnO2 (s), (HARMFUL) – see CLEAPSS Hazcard HC060.
Procedure
- Weigh 6.5 g of lead(II) oxide, 3.5 g of boric acid and 0.5 g of zinc oxide into the boiling tube and stopper firmly, taking great care not to spill any of these chemicals in this process.
- Insert the bung in the tube and shake the contents to ensure thorough mixing, and transfer into the crucible.
- Straighten out a paper clip to form a wire stirrer, and stir the mixture again.
- Place the lid on the crucible, and carefully sit the crucible on the clay pipe triangle which is set-up on a tripod with a heat resistant mat.
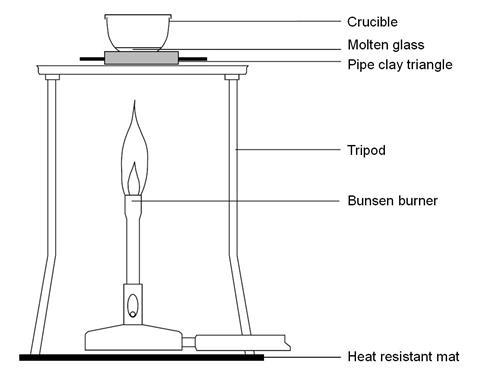
- Heat carefully at first, then strongly with a hot Bunsen flame, until the mixture becomes molten and runny.
- Taking great care, remove the Bunsen flame from underneath the crucible, then use tongs to remove the lid and lift the crucible off the tripod. Pour one or two drops of the molten glass onto the heat resistant mat. Replace the crucible onto the tripod, and keep heating.
- Allow the glass beads to cool on the mat for 5 minutes and then examine them.
- Use the straightened paper clip to pick up a tiny speck of one of the metal oxides provided and stir this into the remaining molten mixture. Do not add too much powder or you will produce a very dark piece of glass.
- Remove the Bunsen flame, and use tongs to pour out one or two drops of the coloured glass from the crucible to form beads on the mat. Note the colour of the glass you have now produced. Place the crucible on the mat to cool.
- Allow all the apparatus to cool before clearing away.
Teaching notes
Very little background knowledge is required, and practical skills are more important in this experiment, including the use of balances and the handling of hot apparatus.
The glass produced is very brittle and difficult to keep. This type of glass is not used commercially, but for further information about the composition of a wide range of glasses made industrially, see links below.
Different groups can be allocated different transition metal oxides to produce coloured glass.
Further information
British Glass provides a very informative introduction to the manufacture of different glasses for different uses.
Additional information
This is a resource from the Practical Chemistry project, developed by the Nuffield Foundation and the Royal Society of Chemistry.
Practical Chemistry activities accompany Practical Physics and Practical Biology.
The experiment is also part of the Royal Society of Chemistry’s Continuing Professional Development course: Chemistry for non-specialists.
© Nuffield Foundation and the Royal Society of Chemistry


















No comments yet