Encourage students to understand nitrogen dioxide better with this experiment, showing them some of its reactions, and putting this gas in an active learning context
In this practical, students will use several different substances in a controlled experiment to explore some of the reactions of nitrogen dioxide. Learning about this important gas, how it’s made, and its use in chemistry.
This practical should take 20 minutes.
Equipment
Apparatus
- Eye protection
- Clear plastic sheet (eg OHP sheet)
- Plastic Petri dish (base + lid), 9 cm
- Plastic pipette
- Scissors
Chemicals
Solutions should be contained in plastic pipettes. See the accompanying guidance on apparatus and techniques for microscale chemistry, which includes instructions for preparing a variety of solutions.
- Nitric acid (concentrated HNO3) diluted 1:1 with water ca 5 M
- Full-range indicator solution diluted 1:1 with deionised water
- Potassium iodide, 0.2 mol dm–3
- Potassium iodate(V), 0.1 mol dm–3
- Potassium bromide, 0.2 mol dm–3
- Potassium bromate(V), 0.1 mol dm–3
- Ammonia solution, 3 mol dm–3
- Copper turnings
Health, safety and technical notes
- Read our standard health and safety guidance.
- Students must wear suitable eye protection (Splash resistant goggles to BS EN166 3).
- Nitrogen dioxide is extremely toxic and corrosive if inhaled, with sometimes delayed effects. It is important to ensure that the amount of NO2 generated does not result in significant leakage from the Petri dish (see CLEAPSS Hazcard HC068b). No more than 2 copper turnings should be used.
- Concentrated nitric acid, HNO3(aq), 5 mol dm–3, is CORROSIVE and gives off toxic fumes (see CLEAPSS Hazcard HC067).
- Potassium iodate(V), KIO3(aq),0.1 mol dm–3, potassium bromate(V), KBrO3(aq), 0.1 mol dm–3 (see CLEAPSS Hazcard HC080), potassium bromide, KBr(aq), 0.2 mol dm–3 and potassium iodide, KI(aq), 0.2 mol dm–3 are low hazard (see CLEAPSS Hazcard HC047b).
- Ammonia solution, NH3(aq), 3 mol dm–3 is corrosive and a respiratory IRRITANT (see CLEAPSS Hazcard HC006).
Procedure
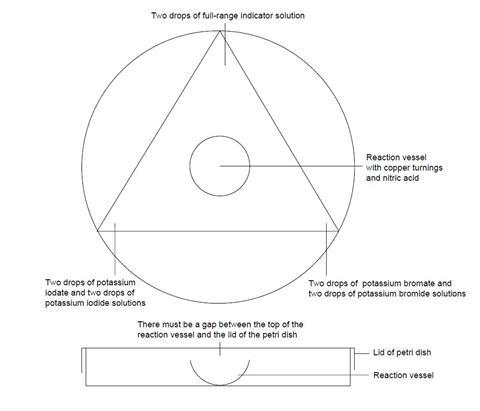
- Cover the worksheet with a clear plastic sheet.
- Place the base of the Petri dish directly over the circle indicated on the worksheet. Place the reaction vessel in the centre of the Petri dish.
- Put two drops of full-range indicator where shown.
- At another corner of the triangle, place two drops of ammonia solution. Place the lid on the Petri dish and wait for the indicator drop to change colour.
- Remove the lid from the Petri dish and, using a piece of tissue, mop up the drop of ammonia.
- At the two remaining corners of the triangle, add the two other test solutions.
- Add a few copper metal turnings to the reaction vessel, followed by three drops of nitric acid. Quickly replace the lid on the Petri dish.
- Record all your observations over the next 15 minutes.
Method
Copper turnings + nitric acid generates first nitric oxide, which then reacts with air to give nitrogen dioxide:
3Cu(s) + 8HNO3(aq) → 3Cu(NO3)2(aq) + 2NO(g) + 4H2O(l)
then: 2NO(g) + O2(g) → 2NO2(g)
Observations
Full-range indicator turns from green to yellow-red indicating that nitrogen dioxide is an acidic gas.
The iodate/iodide solution turns black due to: IO3–(aq) + 5I–(aq) + 6H+(aq) → 3I2(g) + 3H2O(l) also indicating the acidic nature of the gas.
A similar reaction occurs with bromide/bromate.
Student question
- What explanations can you give for your observations?
Downloads
Some reactions of nitrogen dioxide - teacher notes
PDF, Size 0.13 mbSome reactions of nitrogen dioxide - teacher notes
Word, Size 52.22 kbSome reactions of nitrogen dioxide – student sheet
PDF, Size 0.21 mbSome reactions of nitrogen dioxide – student sheet
Word, Size 0.12 mb
References
S. W. Breuer, Microscale practical organic chemistry. Lancaster: Lancaster University, 1991.
Additional information
This resource is part of our Microscale chemistry collection, which brings together smaller-scale experiments to engage your students and explore key chemical ideas. The resources originally appeared in the book Microscale chemistry: experiments in miniature, published by the Royal Society of Chemistry in 1998.
© Royal Society of Chemistry
Health and safety checked, 2018


















No comments yet