Some students do not logically relate the reactivity of a substance to the stability of the chemical species it is comprised of
This activity is inspired by research which suggests that students often believe that atoms will spontaneously form species with octet structures, regardless of the chemical context.
Stability and reactivity questions
The three questions all refer to the diagram. Please select one option for each of the three questions, and explain your reasons.
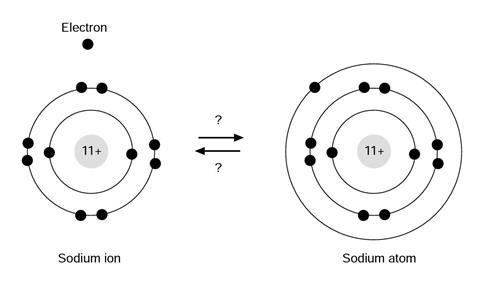
Question 1
Please select one option for the question, and explain your reasons.
- The sodium atom is more stable than the sodium ion.
- The sodium ion is more stable than the sodium atom.
- The sodium ion and sodium atom are equally stable.
- I do not know which statement is correct.
Question 2
Please select one option for the question, and explain your reasons.
- The sodium atom will eject (give out) an electron to become an ion.
- The sodium ion and electron will combine to become an atom.
- Neither of the changes suggested above will occur.
- I do not know which statement is correct.
Question 3
Please select one option for the question, and explain your reasons.
- The sodium atom is more reactive than the sodium ion.
- The sodium ion is more reactive than the sodium atom.
- The sodium ion and sodium atom are equally reactive.
- I do not know which statement is correct.
Answers
In discussing the responses it is suggested that the teacher should focus on the figure on the sheet, and begin by considering question 2. The electron is negatively charged and the ion positive, so they will be spontaneously attracted to form an atom.
The atom will not emit an electron without an input of energy. In the context of the diagram, then, the atom is more stable (question 1), and the ion is more ‘reactive’ (question 3).
It should be borne in mind, however, that this exercise is intended as a probe, and not a test. Some students are likely to think in terms of more familiar chemical contexts, eg comparing a sodium ion in sodium chloride with elemental sodium (which students may consider to contain atoms), where the metal is part of a more reactive system in the presence of water, or air etc.
An important teaching point is that students need to consider the overall chemical context when judging ‘stability’ or ‘reactivity’.
- The sodium atom is more stable than the sodium ion.
- The sodium ion and electron will combine to become an atom.
- The sodium ion is more reactive than the sodium atom.
Notes
For the full version of this chapter, see downloads below.
Downloads
Stability and reactivity
PDF, Size 0.26 mb
Websites
Additional information
These resources have been taken from the book, Chemical Misconceptions : Prevention, diagnosis and care: Theoretical background, Volume 2, by Keith Taber.

Chemical misconceptions

Discover classroom strategies and activities to tackle common misconceptions among students in chemistry, and explore the theory behind different approaches.
- 1
- 2
- 3
- 4
- 5
- 6
- 7
- 8
- 9
- 10
- 11
- 12
- 13
- 14
- 15
- 16
- 17
- 18
- 19
- 20
- 21
- 22
- 23
- 24
- 25
- 26
- 27
- 28
- 29
 Currently
reading
Currently
reading
Stability and reactivity
- 31
- 32
- 33
- 34
- 35
- 36





























































































No comments yet