Observe what happens as ice cubes floating on oil start to melt and the density of the water changes
Although a very safe and simple experiment, this is more likely to be done as a quick teacher demonstration with class discussion, than as a class experiment. Ten minutes should be ample to make the observations, although discussion of these may be as long or as brief as the teacher decides.
Students can observe a variety of interesting surface tension phenomena as the ice begins to melt, producing liquid water with a greater density.
Equipment
Apparatus
- Measuring cylinder, 1 dm3 (see note 2)
- Flexi-cam, webcam or similar to display magnified image of events on suitable screen, if available
Chemicals
- Water, 400 cm3
- Ice cubes (see note 3)
- Cooking oil, 400 cm3, eg pure vegetable oil
Health, safety and technical notes
- Read our standard health and safety guidance.
- A glass measuring cylinder is preferable to a plastic one, to give clear visibility of events.
- The ice cubes should be made from water coloured with a blue food dye to ensure visibility for the class, and well frozen to ensure no liquid water remains trapped inside the cubes. Check that the ice cubes actually float on the oil you intent to use.
Procedure
- Place about 400 cm3 of water and 400 cm3 of cooking oil in the measuring cylinder. Allow the two layers to separate fully; the oil will be on the top.
- Drop an ice cube into the cylinder. It will float (just) on top of the oil.
- Observe the cube. As it melts, the water that is formed makes a droplet attached to the cube. Eventually this detaches itself from the cube and sinks, joining the water layer below. This illustrates the anomalously greater density of water compared to ice.
Teaching notes
Water that has been dyed blue is easily seen in the pale yellow oil against a white background.
A number of other interesting observations can be made, mostly associated with the high surface tension of water:
- After most of the cube has melted, the weight of the water drop is sufficient to drag the remainder of the ice cube down with it; ie the average density of the cube and drop is greater than that of the oil. Sometimes, as the cube and drop are sinking, the drop detaches itself from the cube and the cube floats back to the surface.
- Small mini-droplets occasionally break off from the main one as it descends forming a ‘string of pearls’ effect.
- Water droplets may sit for some time on the water-oil interface without coalescing with the body of the water.
- There are interesting changes of shape in the water droplet as it forms, detaches from the cube, and as it sinks.
- When the coloured water droplets begin to coalesce with the water, the coloured water can be seen to sink as it mixes, because of its greater density.
The demonstration may need to be repeated with an uncoloured ice cube to show that the same density changes occur.
The density of ice is about 0.92 g cm–3 and that of water is about 1.00 g cm–3 at 0 °C. Cooking oil has a density between these two and therefore ice floats on the oil whereas water sinks. Most solids are denser than their liquids. The lower density of ice is caused by its open lattice structure, a hydrogen-bonded, tetrahedral network similar to that of diamond:
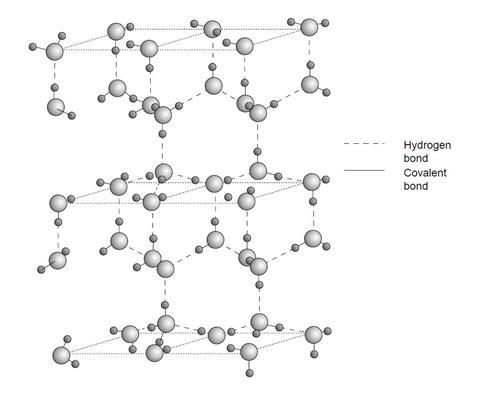
The structure of liquid water has water molecules more closely packed than this below 4 °C, leading to a higher density.
Additional information
This is a resource from the Practical Chemistry project, developed by the Nuffield Foundation and the Royal Society of Chemistry.
Practical Chemistry activities accompany Practical Physics and Practical Biology.
This demonstration is based on an idea developed by Colin Johnson at Techniquest, Cardiff.
© Nuffield Foundation and the Royal Society of Chemistry


















No comments yet