A question and answer sheet to test learner’s knowledge of thermodynamics
The topics covered in this Starter for ten are: important definitions, Born-Haber cycles, enthalpy of solution, entropy, and Gibbs free energy.
Example questions
For each of the transformations represented by the equations 1-5, name the enthalpy change and provide a full definition.
e.g. ½ N2(g) + 3/2 H2(g) → NH3(g)
Name: The standard molar enthalpy change of formation of ammonia
Definition: The enthalpy change when one mole of a compound is formed from its elements under standard conditions, all reactants and products in their standard states
1. O-(g) + 1 e-(g) → O2-(g)
Name:
Definition:
2. K+(g) + aq → K+(aq)
Name:
Definition:
3. NaCl(s) + aq → Na+(aq) + Cl-(aq)
Name:
Definition:
4. Mg2+(g) + O2-(g) → MgO(s)
Name:
Definition:
5. HCl(g) → H+(g) + Cl-(g)
Name:
Definition:
In 1918, a chance meeting of two German scientists, Max Born and Fritz Haber led to a discussion of the manner in which an ionic compound is formed when a metal reacts with a non-metal. This resulted in the creation of what we know today as Born Haber cycles.
The Born Haber cycle for the formation of calcium oxide is shown in the diagram below;
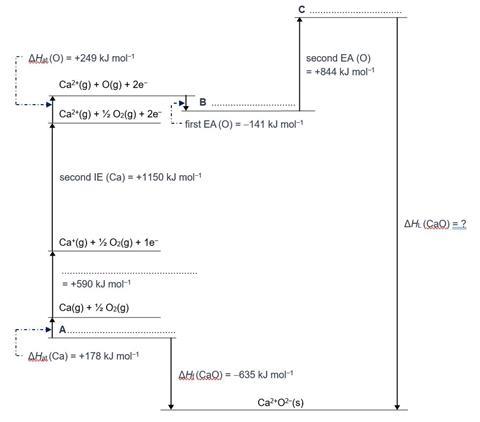
- Complete the cycle by adding suitable labels for the one missing enthalpy change and the missing intermediate species on the three stages labelled A-C.
- Explain why the second electron affinity of oxygen is positive.
- Use the values for the enthalpy changes given to calculate the lattice dissociation enthalpy for calcium oxide.
Notes
This question and answer sheet is available from the ‘Downloads’ section below. an editable version is also available.
Downloads
Thermodynamics
PDF, Size 0.39 mbThermodynamics - editable
Word, Size 0.29 mb
Starters for 10: Advanced level 2 (16–18)

This chapter in our Starters for ten series covers: kinetics, equilibria, acids and bases, carbonyl chemistry, aromatic chemistry, compounds with amine groups, polymers, structure determination, organic synthesis, thermodynamics, periodicity, redox equilibria, transition metal chemistry, and inorganics in aqueous solution.















































No comments yet