Use this demonstration to practise weighing gases to calculate their relative molecular masses using the ideal gas equation
In this experiment, students observe as different gases are weighed using a gas syringe of known volume. The demonstrator begins by weighing the mass of the syringe when ‘empty’, before weighing its mass when filled with a particular gas under known conditions. Students then use the ideal gas equation to calculate the number of moles and hence the relativel molecular mass (RMM) of the gas. Careful manipulation is needed to obtain acceptable results.
This is most likely to be done as a teacher demonstration. Teachers of advanced students may wish to consider the possibility of a student practical for students with good manipulative skills, using appropriate gases.
The preparation of bags containing different gases, and the modification of the syringes, should be done before the lesson, as these procedures may take a considerable time. The demonstration itself should then take about 10 minutes for each gas used.
Equipment
Apparatus
- Eye protection
- Syringe (disposable plastic), 50 cm3 or larger if available, fitted with hypodermic needle (see notes on preparing the syringe below)
- Small rubber bung (exact size unimportant)
- Nail, 5 cm long
- For each gas bag (see notes on creating the gas bags below):
- Polythene bag (sandwich or freezer bag), 1–2 dm3 capacity
- Rubber bung, one hole, 17 mm or similar
- Glass tubing, 4–5 cm length, to fit rubber bung
- Rubber septum cap, self-sealing, to fit the glass tubing
- Cable tie (to fit around the rubber bung)
- Top-pan balance, accurate to +/– 0.001 g, with output to a computer to display readings, if available (an accuracy of +/– 0.01 g is not really sufficient for successful results)
- Thermometer to measure room temperature, accurate to +/– 0.5 ºC, with digital display if available
- Access to an accurate measurement of atmospheric pressure
Chemicals
The experiment requires gas bags filled with various gases:
- Hydrogen
- Oxygen
- Nitrogen
- Methane
- Carbon dioxide
Health, safety and technical notes
- Read our standard health and safety guidance.
- Wear eye protection throughout.
- Remove all possible sources of ignition when handling flammable gases. Ensure adequate room ventilation.
- Hydrogen, H2 (g), (EXTREMELY FLAMMABLE) – see CLEAPSS Hazcard HC048.
- Oxygen, O2 (g), (OXIDISING) – see CLEAPSS Hazcard HC069.
- Nitrogen, N2 (g) – see CLEAPSS Hazcard HC068A.
- Methane (natural gas), CH4 (g), (EXTREMELY FLAMMABLE) – see CLEAPSS Hazcard HC045a.
- Carbon dioxide, CO2 (g) – see CLEAPSS Hazcard HC020a.
Preparing the syringe
The syringe may be of capacity 50 cm3 or 100 cm3, and should be gas-tight when tested with the nozzle blocked with a finger, and have a freely moving plunger. There should be no change in the volume reading if, when it contains 50 cm3 or 100 cm3 of air, the plunger is pushed in and pulled out by 10 cm3 and then released. Rotate the plunger gently before each measurement to ensure the plunger is moving freely within the barrel, and the pressure is equalised inside and outside the syringe.
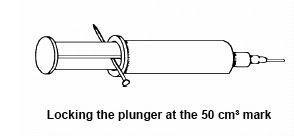
The syringe should be modified as follows (figures for a 50 cm3 capacity syringe):
- Pull out the plunger so the volume of air in the syringe is 50 cm3.
- Warm the nail in the Bunsen flame and push it through the stem of the plunger as shown in the figure, so that the plunger is ‘locked’ at the 50 cm3 mark. This operation should be done in a fume cupboard if the plastic of which the syringe is made is not polythene or polypropene. If in doubt, use a fume cupboard.
Creating the gas bags
Note: wear eye protection.
- When inserting the glass tube through the rubber stopper, use a lubricated cork borer – use care. About 1 cm of tube should protrude from the top of the stopper.
- Gather the neck of the plastic bag around the stopper and secure it tightly with the cable tie. Check for leaks by closing the end of the glass tube with a rubber septum cap, immersing the assembly in a bowl of water, and squeezing.
- Remove the rubber cap and squeeze the air from the bag. It can now be filled with the selected gas from any suitable source, such as a cylinder or chemical generator.
- Fill the bag and squeeze out the gas two or three times to ensure that any air is flushed out. Once finally filled, close the glass tube with the rubber cap. Gas can now be drawn from the bag into the syringe by injecting the hypodermic needle through the cap and sucking gas into the syringe.
The gas bags should be filled shortly before the lesson to minimise any losses by leakage. A selection from the following would be suitable, preferably from gas cylinders or (for methane) the laboratory gas supply (if natural gas):
- Hydrogen (EXTREMELY FLAMMABLE)
- Oxygen (OXIDISING)
- Nitrogen
- Methane (natural gas) (EXTREMELY FLAMMABLE)
- Carbon dioxide
Note
Oxygen and carbon dioxide are also available in small pressurised canisters. However, the purity of the gases in these cylinders, and of methane from the laboratory natural gas supply, may be significantly less than 100%, which will affect the RMM value obtained.
If cylinders are not available, then appropriate chemical generators will have to be used. Refer to the standard techniques for generating, collecting and testing gases to produce:
- Hydrogen, from zinc and dilute sulfuric acid
- Oxygen, from decomposition of hydrogen peroxide catalysed by manganese(IV) oxide
- Carbon dioxide, from calcium carbonate (marble chips) and dilute hydrochloric acid
The gases from these preparations will contain appreciable proportions of water vapour, and will therefore need to be dried appropriately if maximum accuracy is required.
Procedure
- Remove the nail from the syringe and push the plunger in to empty it. Stab the hypodermic needle into the rubber bung to seal the syringe.
- Withdraw the plunger and ‘lock’ it at the 50 cm3 mark with the nail. The syringe now contains 50 cm3 of ‘vacuum’.
- Weigh the syringe, stopper and nail, and note the mass.
- Remove the rubber stopper from the needle and the nail from the hole and push in the syringe plunger completely.
- Insert the needle through the self-sealing stopper of one of the gasbags and suck 50 cm3 of gas into the syringe.
- Seal the gas in by sticking the needle into the rubber bung again, and use the nail to ‘lock’ the syringe. It is not really necessary to lock the syringe in this case, but it does ensure that the nail is weighed along with the syringe.
- Weigh the syringe, bung and nail. The difference between the two masses represents the mass of the gas under investigation.
- Flush out the gas and repeat this procedure with the other gases.
Teaching notes
There are two possible routes for using the results of the experiment to calculate the RMM:
- Use the ideal gas equation PV = nRT to calculate n, the number of moles of gas, which is equal to the mass of the gas in the syringe divided by its molar mass in grams. This requires the recording of room temperature and pressure at the time of the experiment.
- Alternatively use the approximation that one mole of any gas occupies 24 dm3 under average room conditions.
Since the experiment has established the mass of 0.05 dm3 of gas, the mass of 1 mole is simply (24 / 0.05 = 480) times that mass.
Some students may not understand why an empty syringe with the plunger on the zero mark cannot be used for the mass of the syringe with no gas. Be prepared to explain the buoyancy effect of displaced air. The mass of the syringe in this configuration can be measured and compared with its mass with the plunger locked at 50 cm3 to confirm that there is a difference.
Additional information
This is a resource from the Practical Chemistry project, developed by the Nuffield Foundation and the Royal Society of Chemistry. This collection of over 200 practical activities demonstrates a wide range of chemical concepts and processes. Each activity contains comprehensive information for teachers and technicians, including full technical notes and step-by-step procedures. Practical Chemistry activities accompany Practical Physics and Practical Biology.
© Nuffield Foundation and the Royal Society of Chemistry

















No comments yet