Investigate plant water storage crystals as one application of hydrogels in this fun class practical
In this experiment, students use plant water storage crystals to explore the behaviour of hydrogels as polymeric smart materials. After using food colouring or strong tea solution to colour the crystals, students transfer the crystals to different solutions of salt and sugar in distilled water, making detailed observations throughout, noting any changes in the crystals’ colour and volume. The practical work is fun to do, and the results are clear and easy to see.
To complete all parts of this experiment takes over an hour. If lessons are shorter than that then part 1 can be done in a prior lesson. The crystals keep for a few days if they are covered in water.
During the time that the crystals have to be left, other experiments with hydrogels, using hair gel and disposable nappies, could be carried out.
It is a good idea to ask students to make detailed observations of each part of the experiment.
The water crystals can be coloured with a few drops of food colouring (for wonderful, lurid colours), with strong tea solution (which stains some containers but provides a useful model of a drug delivery system – see Teaching notes below) or not at all (which seems a bit of a shame as they look great when coloured).
Equipment
Apparatus
- Eye protection
- Large ice cream tub or similar container, at least 1 dm3
- Beakers, 250 cm3, x3
- Dessert spoon or similar measure
- White paper, to place under beakers to see what is happening more easily
- Stirring rods, x3
- Petri dishes – lids not required, x2
- Access to a sieve (the plastic ones used for sifting flour are fine) or large funnel and either paper towels or filter paper
- Access to a tea strainer (only required if a funnel is used earlier, otherwise the sieve can be used again)
Chemicals
- Water crystals, about 100 cm3 (see note 3 below)
- Either strong tea, 500 cm3, or a few drops of food colouring (optional) (see note 4)
- Sodium chloride (table salt) solution, very concentrated or saturated, 200 cm3
- Distilled water, 400 cm3 (see note 5)
- Sugar, 1 spoonful
Health, safety and technical notes
- Read our standard health and safety guidance.
- Wear eye protection throughout.
- Water crystals are available from garden centres and are sold under various names including Phostrogen Swellgel. Each group needs about a teaspoonful.
- For the strong tea use two tea bags per litre, pour on boiling water and leave to brew overnight. This tea stains some containers.
- If distilled water is not available, tap water can be used but the results are not as spectacular.
Procedure
Part 1
- Estimate the volume of one teaspoonful of the water crystals.
- Put about 500 cm3 of tea, tap water or water coloured with a few drops of food colouring into the beaker or tub. Add one teaspoonful of water crystals, stir gently and leave on one side for at least half an hour, or overnight.
Part 2
- Sieve the water crystal mixture. It is best to do this over a large tub rather than the sink in case you drop it. Wash the gel crystals carefully once or twice in water to remove any excess tea or food colouring if you used it. Estimate the new volume of your crystals.
- Stand the three 250 cm3 beakers on a piece of white paper.
- Put two dessert spoons of the gel crystals into each beaker, estimate their volume and then add about 200 cm3 of salt solution to one and 200 cm3 of distilled water to each of the others. Add a spoonful of sugar to one of the beakers with water in it. Label the beakers.
- Stir the mixtures gently – using a separate stirring rod for each one so that the solutions do not become cross-contaminated. Leave for 10–15 minutes, stirring occasionally.
- If you used tea, pour some of the solution from each beaker into a petri dish placed on the white paper. Use a tea strainer or sieve to prevent any crystals getting onto the petri dish. Note carefully the colour of each liquid.
- Sieve the remaining mixtures, discarding the excess liquid and returning the crystals to the beakers. Estimate their new volumes.
Teaching notes
This activity can be used to enhance the teaching of ionic and covalent bonding, or hydrogels can be considered as an interesting polymer as well as an example of a smart material. Hydrogels are smart materials because they change shape when there is a change in their environment – in this case it is the change in the concentration of ions.
Students need to have some knowledge and understanding of ionic and covalent bonding, reversible reactions, and acids and bases to understand what is happening.
Hydrogels are polymers that can retain many times their own weight in water. They are often polymers of carboxylic acids that ionise in water, leaving the polymer with several negative charges down its length. This has two effects. First, the negative charges repel each other and the polymer is forced to expand. Secondly, polar water molecules are attracted to the negative charges. This increases the viscosity of the resulting mixture still further as the polymer chain now takes up more space and resists the flow of the solvent molecules around it.
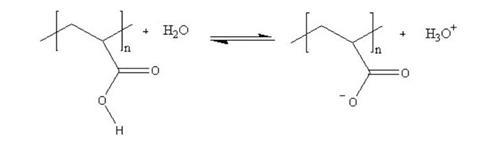
The polymer is in equilibrium with the water around it, but that equilibrium can be disturbed in a number of ways. If the the ionic concentration of the solution is increased – eg by adding salt – the positive ions attach themselves to the negative sites on the polymer, effectively neutralising the charges. This causes the polymer to collapse in on itself again. Adding alkali removes the acid ions and moves the equilibrium to the right; adding acid has the opposite effect.
There are a large number of hydrogels and they are sensitive to different pHs, temperatures and ionic concentrations. By using a mix of monomers to create the polymer these characteristics can be fine-tuned.
The hydrogels that are commonly available and are used in this practical activity are sensitive to salt concentration, but do not show much change across the pH range that can be readily investigated in the classroom. However, they do lend themselves very well to a range of investigative practical work. For example, their volume in different amounts of water or in different salt concentrations can be measured. For this type of investigation it is best to use either plant water crystals or to order sodium polyacrylate from Sigma Aldrich – this has a smaller crystal size and gives faster results.
Students should make detailed notes on their experiments, noting changes in volume, colour and any other observations. Some expected observations could include:
- The crystals swell up from about 5 cm3 to about 500–600 cm3. They take on the colour of the tea (or food colouring), showing that the tea has also been absorbed.
- When distilled water is added to the hydrated crystals, they swell up further. The tea remains absorbed in the crystals and the water does not change colour.
- When salt water is added to the hydrated crystals, they begin to shrink and the water changes colour as the tea is released. It is possible to measure the aproximate size of individual pieces of the hydrogel too, and to show that the pieces have swollen or shrunk.
- The hydrated crystals in the sugar solution have the same volume as the ones in the distilled water. If they are left for up to 15 minutes the tea is not released. (After this time, the water in the hydrated crystals is in equilibrium with the water in the beaker and some tea may begin to be observed.)
These observations show that the hydrogel responds to changes in the ionic concentration of the solution – the salt, which is ionic, causes the hydrogel to collapse but the covalent sugar does not.
Research is currently being done to see if it is possible to use hydrogels and similar materials as a drug delivery system – a way to get drugs and medicines to where they are required in the body. The experiment with tea and the hydrogel is a model of this type of drug delivery system. The drug is first loaded onto the carrier and then it is released at the right location. The tea represents the drug and the hydrogel is the carrier. Use the worksheet available for download below to explore this model in more detail.
Downloads
Drug delivery and smart materials worksheet
Editable handout | Word, Size 0.77 mbDrug delivery and smart materials worksheet
Handout | PDF, Size 0.84 mb
Additional information
This is a resource from the Practical Chemistry project, developed by the Nuffield Foundation and the Royal Society of Chemistry.
Practical Chemistry activities accompany Practical Physics and Practical Biology.
The experiment is also part of the Royal Society of Chemistry’s Continuing Professional Development course: Chemistry for non-specialists.
© Nuffield Foundation and the Royal Society of Chemistry


















No comments yet