Clean up a microscale oil spill in your classroom, and look deeper into the impact of this destructive environmental event
In this experiment, students will learn how oil spills are addressed, using a micro version of the large scale event. Explore the chemistry behind a very real natural disaster, and look deeper into its advantages and disadvantages.
This experiment should take 10 minutes.
Equipment
Apparatus
- Eye protection
- Student worksheet
- Beaker, 100 cm3
- Plastic pipette
- Scissors
Chemicals
- Soil-moist hydrocarbon polymer (see notes section for more information)
- Oil or paraffin
Health, safety and technical notes
- Read our standard health and safety guidance.
- Students must wear suitable eye protection (splash proof goggles to BS EN166 3).
- Avoid inhaling fumes.
- Paraffin is FLAMMABLE, ensure no naked flames or other sources of ignition (see CLEAPSS Hazcard HC045b).
Procedure
- Half-fill a 100 cm3 beaker with water.
- Using your pipette, add some oil or paraffin to the beaker to give a thin layer on top of the water.
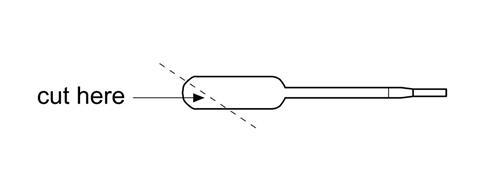
- Cut off the end of a pipette to form a scoop as shown below.
- Add two scoops of polymer powder to the beaker and stir with the end of the pipette.
Observations
On adding the polymer, and stirring, a rubbery solid is formed very quickly and the layer of oil/ paraffin disappears.
Student questions
- What do you observe?
- How do you explain your observations?
- If you were to do this experiment on a large scale to try to deal with an oil slick at sea, what would be the advantages of using this polymer powder and what difficulties might you encounter?
Notes
The essential ingredient in this experiment is the powdered polymer, which can be obtained from Flinn Scientific produce (Enviro-Bond 403) at £8 for 100 g.
With careful use, 30 g should provide enough for many experiments.
The polymer itself is a copolymer of acrylamide and hydroxymethylmethacrylate, crosslinked and dehydrated.
A similar substance is produced commercially by BP under the tradename Rigidoil.
Downloads
The treatment of oil spills - student sheet
PDF, Size 0.12 mbThe treatment of oil spills - student sheet
Word, Size 63.55 kbThe treatment of oil spills - teacher notes
PDF, Size 0.13 mbThe treatment of oil spills - teacher notes
Word, Size 51.73 kb
References
S. W. Breuer, Microscale practical organic chemistry. Lancaster: Lancaster University, 1991.
Additional information
This resource is part of our Microscale chemistry collection, which brings together smaller-scale experiments to engage your students and explore key chemical ideas. The resources originally appeared in the book Microscale chemistry: experiments in miniature, published by the Royal Society of Chemistry in 1998.
© Royal Society of Chemistry
Health and safety checked, 2018

















No comments yet