Quiz your students on kinetics topics
The topics included in this Starter for ten activity are: collision theory, sketching Maxwell-Boltzmann and the importance of Maxwell-Boltzmann.
Example questions
This question is all about the reaction between zinc metal and hydrochloric acid to produce zinc chloride and hydrogen gas.
- Write a balanced symbol equation for the reaction that occurs.
- The reaction flasks below show the same reaction but under different conditions. The acid is in excess in all five flasks.
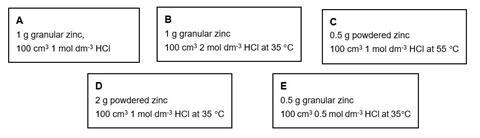
- In which flask is the reaction rate the slowest?
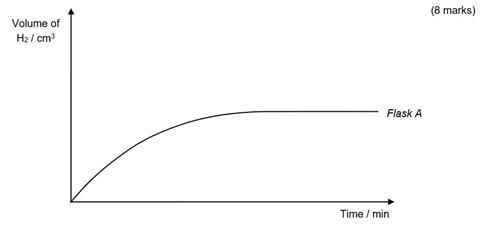
- The graph below shows how the volume of hydrogen given off changed with time for the reaction that occurred in flask A. Sketch on the same set of axes, the curves you would expect to get if you repeated the measurements for flasks B, C, D and E.
Notes
A full version of this question and answer sheet is available from the ‘Downloads’ section below. An editable version is also available.
Downloads
Kinetics
PDF, Size 0.31 mbKinetics - editable
Word, Size 0.22 mb
Starters for 10: Advanced level 1 (16–18)

This chapter in our Starters for ten series covers quantitative chemistry, atomic structure, bonding, trends in the periodic table, organic chemistry, thermodynamics, kinetics, equilibria, redox, analysis and experimental skills.
- 1
- 2
- 3
- 4
- 5
- 6
- 7
 Currently
reading
Currently
reading
Kinetics
- 9
- 10
- 11
- 12









































No comments yet