Quiz your students on their redox knowledge
The topics covered in this Starter for ten activity are: oxidation numbers, writing half equations, half equations to overall equations, electrolysis of aluminium, extraction of other metals, displacement reactions, ability to reduce sulfuric acid, uses of chlorine and chlorate (I), halogens summary.
Example questions
Work out the oxidation numbers for the bold elements in the compounds and perform the calculation.
Present your answer to your teacher. There is 1 mark for each correct oxidation number you have deduced.
Note: You are calculating the oxidation number of the element, not its overall contribution to the compound eg, CaCl2 you would give the answer for Cl as -1, not as …Cl2 (-2).
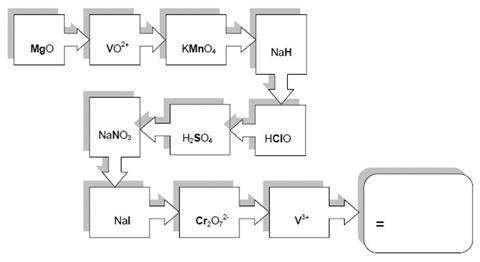
Balance the half equations by balancing the atoms and adding
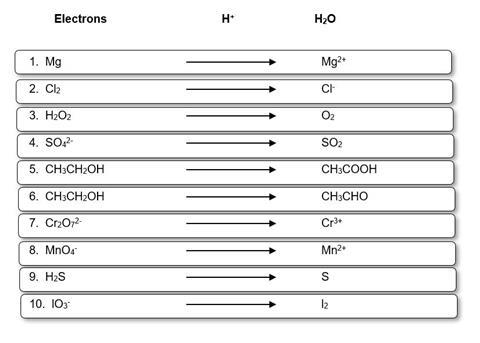
Notes
The full question and answer sheet is available from the ‘Downloads’ section below. An editable version is also available.
Starters for 10: Advanced level 1 (16–18)

This chapter in our Starters for ten series covers quantitative chemistry, atomic structure, bonding, trends in the periodic table, organic chemistry, thermodynamics, kinetics, equilibria, redox, analysis and experimental skills.










































No comments yet