This experiment helps students to understand the way that hydroxybenzene (phenol) reacts with other chemicals, by letting learners create five different reactions within a Petri dish
In this experiment, students will use a Petri dish to create some chemicals reactions of hydroxybenzene (phenol), taking their observations and interpreting the data to understand what is taking place.
This experiment should take 20 minutes.
Equipment
Apparatus
- Eye protection
- Clear plastic sheet (eg OHP sheet)
- Plastic Petri dish, 9 cm diameter
Chemicals
Solutions should be contained in plastic pipettes. See the accompanying guidance on apparatus and techniques for microscale chemistry, which includes instructions for preparing a variety of solutions.
- Deionised water
- Full-range indicator solution – diluted 1:1 with deionised water
- Nitric acid,1 mol dm–3
- Iron(III) nitrate, 0.1 mol dm–3
- Sodium carbonate, 1 mol dm–3
- Sodium hydroxide, 1 mol dm–3
- Hydrochloric acid, 1 mol dm–3
- Hydroxybenzene (phenol)
Health, safety and technical notes
- Read our standard health and safety guidance.
- Students must wear suitable eye protection (Splash resistant goggles to BS EN166 3).
- The students should sample the bottle of hydroxybenzene (phenol) using the method in our guidance on methods and techniques.
- Hydroxybenzene (phenol) is hazardous and gloves should be worn (see CLEAPSS Hazcard HC070a).
- Nitric acid, 1 mol dm–3 HNO3 (aq), is CORROSIVE (see CLEAPSS Hazcard HC067).
- Sodium hydroxide,1 mol dm–3 is corrosive, iron(III) nitrate, Fe(NO3)3.9H2 O (aq), 0.2 mol dm–3, sodium carbonate 1 mol dm–3 and hydrochloric acid, HCl(aq), 1 mol dm–3, are of low hazard (see CLEAPSS Hazcard HC091a, HC055c, HC095a, HC047a).
- Hydroxybenzene (phenol) is CORROSIVE, TOXIC by all routes and causes damage to organs on repeated or prolonged exposure (see CLEAPSS Hazcard HC070a). Dangerous if in contact with sodium nitrate (see CLEAPSS Hazcard HC093).
- Depending on the formulation of the universal indicator, a 1:1 dilution with water might still be flammable. Keep away from sources of ignition.
Procedure
- Cover the worksheet with a clear plastic sheet.
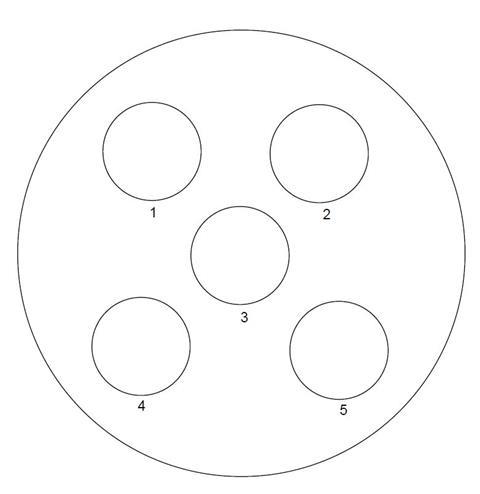
- Place the base of the Petri dish over the circle (below). Using the hydroxybenzene (phenol) sampling procedure (ask your teacher) place small quantities of hydroxybenzene (phenol) in the Petri dish over each of the five small circles.
- In circle 1 add two drops of water, leave for 1 minute then add one drop of full-range indicator solution.
- In circle 2 add two drops of 1 mol dm–3 nitric acid. Observe any changes over the next 5 min.
- In circle 3 add two drops of iron(III) solution.
- In circle 4 add two drops of sodium carbonate solution. Bearing in mind any conclusions that you arrived at in observing circle 1 what might you be looking for here?
- In circle 5 add two drops of sodium hydroxide solution. Observe closely over the next minute. Add one drop of hydrochloric acid and observe closely.
- When you have finished, add drops of sodium hydroxide to the Petri dish to dissolve the hydroxybenzene (phenol) and then mop up with tissue.
Observations
- Hydroxybenzene (phenol) is partially soluble in water, and oily drops should be observed. An acidic solution is formed, and the indicator solution should turn red.
- The mixture turns dark as the hydroxybenzene (phenol) reacts with the nitric acid to give a mixture of nitrophenols.
- A violet colouration is seen, which is characteristic of the reaction between iron(III) and phenolic OH groups.
- No reaction is observed with sodium carbonate solution, indicating that hydroxybenzene (phenol), although acidic, is not a strong enough acid to liberate carbon dioxide from carbonates.
- Hydroxybenzene (phenol) dissolves readily in sodium hydroxide to form sodium phenoxide. The hydroxybenzene (phenol) is liberated and oily drops should be observed when this solution is acidified with hydrochloric acid.
Student questions
- Can you give explanations for your observation?
- Can you write equations for the reactions you have observed?
Downloads
The chemical properties of hydroxybenzene – student sheet
PDF, Size 0.17 mbThe chemical properties of hydroxybenzene – student sheet
Word, Size 95.94 kbThe chemical properties of hydroxybenzene– teacher notes
PDF, Size 0.14 mbThe chemical properties of hydroxybenzene– teacher notes
Word, Size 52.39 kb
References
S. W. Breuer, Microscale practical organic chemistry. Lancaster: Lancaster University, 1991.
Additional information
This resource is part of our Microscale chemistry collection, which brings together smaller-scale experiments to engage your students and explore key chemical ideas. The resources originally appeared in the book Microscale chemistry: experiments in miniature, published by the Royal Society of Chemistry in 1998.
© Royal Society of Chemistry
Health and safety checked, 2018


















No comments yet