Explore simple covalent molecules and hydrocarbons with this series of activities
The resource should take approximately three hours to complete in full. Use it in a sequence of timetabled lessons, science clubs or during an activity day. It was initially created for 14–16 year-old learners, but could be adapted to support revision or transition for learners at the next stage.
Use the resource in a sequence of timetabled chemistry lessons, science clubs or during an activity day.
Learning objectives
- Explain how a covalent bond is formed between two atoms.
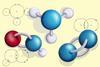
- Construct models for at least three hydrocarbons.
- Draw the displayed structures for three alkanes and three alkenes.
- Work out the structures for at least two additional functional groups and draw their displayed formulas.
Guidance notes
Use the PowerPoint slides to revise covalent bonding, using carbon and hydrogen as examples. Conduct Activities 1 and 2 on hydrocarbons in small groups to find out what learners know at the start of the day, recapping key definitions and structures.
Activity 3 requires molymod kits. Demonstrate how the molymod kit works by making a model of a hydrogen atom. Make the link between the molecular formula, structural formula and molymod model while showing slide 12. Check that learners know how to fill in the table in the student workbook before making their own models of simple molecules. Remind learners how to draw a 2D displayed formula representation before they start.
Introduce the idea of alkanes and relate this back to crude oil and hydrocarbons at the beginning of Activity 4. In small groups, learners will build models of the first four alkanes and draw their displayed formulas. Ask them to identify and use the pattern as they increase the carbon and hydrogen atoms.
Learners should now be able to work out the general formula for an alkane, although some may need extra support here. They can then use the general formula to predict the formulas for pentane, hexane, heptane and octane. Ask them to write the molecular and structural formulas and build models to check their predictions. Encourage learners to connect carbon atoms in a straight line first to avoid isomerism.
Use slide 34 to discuss reactions of alkanes, including complete and incomplete combustion. Activity 5 is a series of demonstrations. Give learners time to record their observations as you show how to use a Bunsen burner, a controlled explosion of methane and how to produce methane bubbles.
Use the introductory key points for Activity 6 in the PowerPoint slides to explain what alkenes are. Task learners to label the double bond in an alkene and then make models of alkenes using the molymod kits. You could introduce isomers to learners at this point and show them some of the other possible structures, but they do not need to understand the concept at this level.

This resource was developed as part of the Chemistry for All project. The project was set up to explore and address barriers to participation in UK chemistry undergraduate study through a longitudinal project. Read the findings relevant to teachers, outreach providers, education policymakers and parents in the summary report, or download the full research report.
Run Activity 7 either as a class practical or demonstration of how alkenes decolourise bromine water. Make sure learners follow all practical procedures and safety instructions completely. Use the PowerPoint to summarise and discuss the learners’ findings.
Learners should now be quite confident using the molymod kits with reference to the structural formulas given. The term functional group may be new to some learners so introduce the concept before Activity 8. In this activity, ask learners to build models of molecules with different functional groups and identify each group by circling them on their drawn displayed formulas. The PowerPoint slides can help to introduce a range of compounds.
The Quick quiz at the end of the PowerPoint recaps some of the key concepts. Use the questions to check for understanding, with learners answering on mini whiteboards, orally in small groups or by whole-class discussion.
Give the Homework questions to learners to complete after the session.
Find the answers to all the activities in the teacher notes and on the slides.
Careers
The Chemistry for All project found that recognising the value and importance of chemistry, and appreciating how it can lead to interesting and well-paid jobs strongly related to learners’ future aspirations.
Use the job profiles embedded in the PowerPoint to introduce learners to four scientists who use their chemistry skills, knowledge and qualifications in their careers. Share our careers website, A Future in Chemistry, with learners to discover the different study options available to them, find more job profiles of chemists making a difference and try our career options game.
Health and safety
Read our health and safety guidance and carry out a risk assessment before running any live practical.
The safety equipment suggested is in line with CLEAPSS requirements. For non-hazardous substances, wearing lab coats can help to protect clothes. The safety rules might be different where you live so it is worth checking local and school guidance.
Find more projects like this, plus shorter activities suitable for single science lessons or clubs on our Outreach resources hub.
Downloads
Bonding workshop student workbook
Editable handout | Word, Size 1.79 mbBonding workshop student workbook
Handout | PDF, Size 0.86 mbBonding workshop teacher notes
Editable handout | Word, Size 1.53 mbBonding workshop teacher notes
Handout | PDF, Size 1.02 mbBonding workshop technician notes
Editable handout | Word, Size 1.6 mbBonding workshop technician notes
Handout | PDF, Size 0.5 mbBonding workshop presentation slides
Presentation | PowerPoint, Size 2.75 mbBonding workshop presentation slides
Presentation | PDF, Size 2.43 mb
Additional information
This resource was originally developed by Liverpool John Moores University to support outreach work delivered as part of the Chemistry for All project
Chemistry for All project resources

Practical resources used by some of the activity providers for outreach work as part of the Chemistry for All project
- 1
- 2
- 3
- 4
- 5
 Currently
reading
Currently
reading
Bonding workshop










































No comments yet