Identify and address misconceptions learners hold about the properties of particles vs the bulk properties of substances
Ask learners to decide if the statements about iron (a familiar example of a metal) on the student sheet are true or false.
-

Download this
A true or false worksheet with teacher guidance and answers to help identify and address any misconceptions learners may hold about metallic bonding.
View and download more Chemical misconceptions resources
How to use this resource
Learners can struggle with using atomic/molecular level models of matter to explain the properties of substances (read more about learners’ beliefs in alternative ideas). It is also common for learners to begin their post-16 studies believing that all materials have either covalent or ionic bonding and that ionic and metallic materials are molecular (read more about learners’ misconceptions related to chemical bonding).
- When to use? Use with learners in the 14–16 age range who have been introduced to the properties of metals and to the basic notion of metallic bonding.
- Group size? Instruct learners to do the task independently in class so you can identify misconceptions.
- Topics assessed? Metallic structure and bonding; explaining properties using the particle model.
- How long? 10–15 minutes
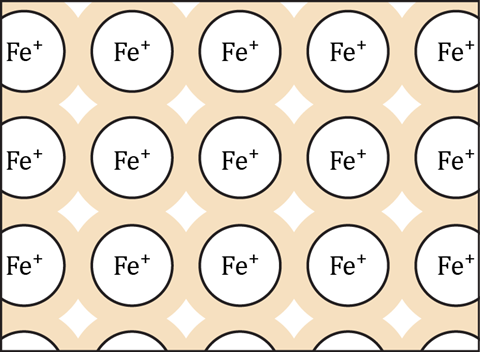
Emphasise to learners that the diagram above shows just a small part of a slice through the lattice structure and that the real structure is three-dimensional. Check that learners understand that the shading in the diagram represents delocalised electrons.
-

Lesson plan available
Build a lesson around a shortened version of this activity with our Metallic bonding and the structure of iron lesson plan for 14-16 year olds. Learners work in groups to challenge misconceptions using ten true or false questions from this diagnostic probe.
Answers
- Iron has a type of bonding called metallic bonding.
TRUE: Iron is a metal, and all metals have a type of bonding called metallic bonding which is different from covalent and ionic bonding. In metallic bonding the outer shells of adjacent atoms overlap, and the outer shell electrons are free to move about through the lattice. The metal consists of metal cations and a balancing number of these ‘free’ electrons. - Iron atoms do not have a full outer shell of electrons, and this makes iron very reactive.
FALSE: Although an isolated iron atom does not have a full outer shell, the outer electrons are involved in the bonding in the metal. Iron is not very reactive, although it will slowly rust. - An iron atom is a silver-grey colour, and so iron metal is a silver-grey colour.
FALSE: The colour of iron is a property of the arrangement of positive ions and electrons. A single atom of iron would not have a colour. - Iron can conduct electricity because some of the iron atoms can slip over their neighbours and move through the solid.
FALSE: The positive iron ions are normally fixed in their lattice positions. It is the electrons from the outer shells that are able to move about, allowing electrical current to flow through the metal. - Iron can be reshaped, without changing the shape of iron atoms.
TRUE: Metals can be worked into different shapes by hammering to force the positive ions to slip over each other. The ions change position, but not shape. - Iron rusts because iron atoms will rust if exposed to damp air.
FALSE: The rusting of iron is due to a chemical reaction between the iron and oxygen and water vapour in the air. During these reactions some of the iron becomes part of a red chemical compound (the rust), but the atoms themselves do not corrode. - In iron metal each atom is bonded to each of the other iron atoms surrounding it.
TRUE: The iron atoms are packed together so that each positive iron ion is surrounded by eight others as if it is in the centre of a cube. The structure is held together by metallic bonding. - Iron conducts electricity because iron atoms are electrical conductors.
FALSE: The metal conducts because the outer shell electrons are able to move through the metallic lattice structure. The individual atoms do not conduct. - Iron is a solid because that is the natural state for metals.
FALSE: The state of a metal depends on the temperature. Deep in the earth, where it is very hot, iron is a liquid. One metal called mercury is a liquid at room temperature. - A metal such as iron consists of positive metal ions, and negative electrons which move around the solid between the ions.
TRUE: The iron structure contains positive iron ions surrounded by fast-moving delocalised electrons. Sometimes this is called a ‘sea of electrons’. - An iron atom will reflect light, and so freshly polished iron shines.
FALSE: Polished metal will form a mirror because of the regular lattice of positive ions and the ‘sea’ of electrons. Individual iron atoms would not reflect light. - Iron becomes a liquid when heated because the bonds melt.
FALSE: The metal melts when enough energy is provided to allow the positive ions to slip over each other. The bonds in the liquid metal are weaker than in a solid metal. If the liquid was heated until it boiled the bonds would break (but not ‘melt’). - Iron conducts electricity because it contains a ‘sea’ of electrons.
TRUE: The electrons are able to move about, and will pass along the metal when it is connected to a battery. - The atoms in a metal such as iron are held together by ionic bonds.
FALSE: The bonding in a metal is metallic bonding. This is different from ionic bonding as there are no negative ions present. - The reason iron conducts heat is because there is room between the atoms for hot air to move through the metal.
FALSE: The positive iron ions are held close together by the metallic bonding, and there is no room for other atoms and molecules to get between them. Heat passes along the metal due to lattice vibrations and the movement of electrons. - The reason that iron is hard is because iron atoms are hard.
FALSE: Hardness is a property of the metal due to the strong bonding holding the structure together. It is the arrangement of positive ions and free electrons which makes the metal hard. - In iron there are molecules held together by magnetism.
FALSE: There are no molecules in a metal - each positive ion is bonded to all those around it by the ‘sea’ of electrons, and those ions are bonded to others, and so on. Each ion in a metallic crystal is bonded (indirectly) to all the others. - If iron is heated to a very high temperature it would become a gas.
TRUE: If a solid metal is heated it will melt, and if heating is continued to a high enough temperature the liquid metal will boil. The boiling point of iron is 2861°C. - Metals such as iron expand when heated because the atoms get bigger.
FALSE: When the metal is heated the positive ions vibrate more, and move a little further apart. - Chemical bonds are needed to hold the atoms together in a metal such as iron, even though all of the atoms are of the same type.
TRUE: The atoms would not remain joined together if there was no bonding between them. This is true for all solids, whether the atoms are of one type (in an element) or different types (in a compound).
Downloads
Iron - a metal (true or false) student sheet
Handout | PDF, Size 0.18 mbIron - a metal (true or false) teacher guidance
Handout | PDF, Size 0.2 mbIron - a metal (true or false) student sheet
Editable handout | Word, Size 0.48 mbIron - a metal (true or false) teacher guidance
Editable handout | Word, Size 0.51 mb
Websites
Additional information
These resources have been taken from the book Chemical Misconceptions: Prevention, Diagnosis and Cure: Theoretical Background, Volume 2, by Keith Taber.
The resource was adapted and updated in 2025 by Kirsty Patterson.

Chemical misconceptions

Discover classroom strategies and activities to tackle common misconceptions among students in chemistry, and explore the theory behind different approaches.
- 1
- 2
- 3
- 4
- 5
- 6
- 7
- 8
- 9
- 10
- 11
- 12
- 13
- 14
- 15
- 16
- 17
- 18
- 19
- 20
- 21
 Currently
reading
Currently
reading
Iron – a metal: true or false?
- 23
- 24
- 25
- 26
- 27
- 28
- 29
- 30
- 31
- 32
- 33
- 34
- 35
- 36














































































































No comments yet